The Pondeville Group

Interplays between physiology and arbovirus infection in mosquito vectors
Male and female mosquitoes feed on plant nectar to get sugars, which are required for their energy and survival. In addition, females from many mosquito species, such as Aedes aegypti, must get a blood meal from a vertebrate host to obtain the necessary proteins to develop their eggs. This requirement for a blood meal results in Ae. aegypti being a vector of numerous arthropod-borne viruses (arboviruses; e.g. dengue [DENV], chikungunya [CHIKV], Zika [ZIKV] viruses), which are global public health threats and economic burdens.
Dr Pondeville’s lab seek to identify the factors that shape mosquito vector competence and how they can impact on global pathogen transmission. Using the CVR state-of-the-art CL2 and CL3 insectaries as well as modern transgenic facilities, we investigate how the mosquito life traits and physiology influence arbovirus infection, mosquito tissue-specific antiviral responses and further transmission.
Our research is critical for both the fundamental understanding of disease emergence and the development and application of vector control strategies to block arbovirus transmission.
Watch a film about the CVR Insectaries
Current Research Projects
Analysing how mosquito life traits and physiology influences antiviral immunity and vector competence
We are analysing the influence of sugar and blood feeding on antiviral immunity and vector competence.
Image: Aedes aegypti females fed with blue-coloured sugar (left) or not (right).

Characterising the importance of the interrelationships between microbiota, nutrition and host physiology
We investigate how the mosquito microbiome influences host nutrition and metabolism and how this in turn affects physiology (immunity, reproduction) and vector competence in the mosquito vector Ae. aegypti.
Image: Mosquitoes host communities of microbes in their digestive tract, which influences various aspects of their biology including vector competence.
MOSAICS, a MOSquito Arbovirus omICS atlas to unravel mosquito-arbovirus interactions
We are currently developing a mosquito arbovirus omics atlas (MOSAICS) to get insights into organs’ response and contribution to whole body changes, response to blood meal and virus infection, and response to infection and dissemination. This collaborative work will provide to the vector research community a rich resource for investigations not previously possible. Collaboration with Buchon’s lab (Cornell University, US).
Assessing the risk of mosquito-borne diseases in Scotland
This 3-year project, funded by the UKRI-DEFRA One Health Approaches to Vector-borne Diseases programme, is a multi-institutional collaboration between investigators at the University of Glasgow (UoG), UK Health Security Agency (UKHSA) and the UK Centre for Ecology and Hydrology (UKCEH), with the aim of understanding the risk of mosquito-borne disease establishment in Scotland under current and future environmental scenarios. It involves a multidisciplinary programme of work spanning mosquito ecology, surveillance of vector-borne pathogens in avian reservoirs and mosquitoes, vector competence studies, ecological and epidemiological modelling. We are partnering with Public Health Scotland and the Scottish Government with the aim of filling evidence gaps to enable national risk assessment and policy development. We are collaborating with stakeholders from environmental agencies in Scotland including Nature Scot, the Royal Society for the Protection of Birds, Forestry and Land Scotland, the Wetland and Wildfowl Trust, the Scottish Wildlife Trust and local councils.

Follow us on Twitter: @MosquitoScot
Developing mosquito genetic tools to study mosquito-arbovirus interactions
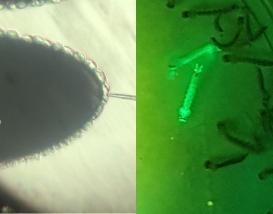
We establish transgenic mosquitoes using site-specific transformation techniques and develop genetic tools such as the Gal4/UAS system to address gene function at the tissue level.
Image: Microinjection of mosquito eggs (left) and GFP transgenic larvae (right).
Identifying and characterizing mosquito genes involved in vector competence
In collaboration with other CVR research groups, we are identifing mosquito genes involved in arbovirus infection in vitro and further characterising them in vivo.
Image: Four identical confocal images symmetrically organized showing the female mosquito midgut, an epithelium (cell nuclei in blue) surrounded by visceral muscles (green). The protein p400 (stained in red) is expressed in tracheae, the respiratory system of insects. Credit: Former PhD student Floriane Almire (Awarded by 3I’S IMAGING COMPETITION).

Determining the roles of arboviral proteins during mosquito infection
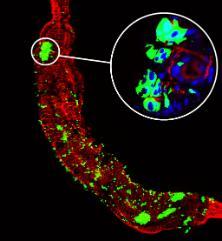
In collaboration with other CVR members, we are investigating the role of bunyamwera NS proteins during mosquito infection.
Image: Mosquito gut infected with BUNV (green).
Examining how mosquito saliva-derived factors influence arbovirus infection in mammals
In collaboration with Dr McKimmie (University of Leeds), we characterise how mosquito biting and saliva enhances infection with a broad range of mosquito-borne viruses.
Interested in joining the laboratory for projects, PhD studies? Email emilie.pondeville@glasgow.ac.uk
Research group members
|
Dr Jean-Philippe Parvy |
Dr Selim Terhzaz |
David Kerrigan |
Collaborators
Internal
External
- Prof Heather Ferguson, University of Glasgow, United Kingdom
- Prof Alain Kohl, LSTM, United Kingdom
- Dr Clive McKimmie, University of Leeds, United Kingdom
- Dr Nicolas Buchon, Cornell University, US
- Dr Julie Reveillaud, INRA, France
- Dr Mathilde Gendrin, Institut Pasteur, French Guiana
- Dr Anna-Bella Failloux, Institut Pasteur, France
- Dr Eric Marois, University of Strasbourg, France
Grants and Awards
Grants and Awards listed are those received whilst working with the University of Glasgow
Impact of immunity and infection on mosquito fitness
MRC Quinquennial Core Funds to CVR
2023-2028
Assessing the risk of insect vector-borne diseases in Scotland and their response to environmental change.
UK Research and Innovation
2023 - 2026
MOSAICS - A mosquito arbovirus omics atlas to unravel mosquito-arbovirus interactions
Biotechnology and Biological Sciences Research Council
2022 - 2023
INFRAVEC-2
European Commission
2017 - 2021
Zikalliance
European Commission
2016 – 2019
Arbovirus interactions with arthropod hosts
MRC Quinquennial Core Funds to CVR
2016-2023





