SPIRIT|CONSORT-SURROGATE

This project aims to develop consensus-driven reporting guidelines for Randomised Controlled Trials and protocols that use surrogate primary endpoints. This will be achieved by developing consensus-driven extensions to the SPIRIT and CONSORT checklists: SPIRIT-SURROGATE and CONSORT-SURROGATE (see lay summary on the Patient and Public Involvement page).
Well designed, conducted, and reported randomised controlled trials (RCTs) provide rigorous scientific evidence for evaluating health interventions. However, RCTs can be time and resource intensive. To make them more time and resource efficient, surrogate endpoints are used as substitutes for patient relevant outcomes (e.g., hospitalisation, death). Traditionally, surrogate endpoint use has focused on the regulatory setting for pharmaceuticals and whether biomarkers are “likely to predict” patient-centred outcomes of interest (e.g., systolic blood pressure for cardiovascular events, low density lipoprotein cholesterol for myocardial infarction, and HIV viral load for development of AIDS). Nevertheless, it is important to acknowledge a more wider application in RCTs of intermediate outcomes that are believed to capture the causal pathway through which pharmaceutical, surgical, organizational or public health interventions impact the ultimate patient-relevant outcome: for example, hospice enrolment for mortality with an intervention aimed at improving end of life care or fruit and vegetable consumption for cardiovascular events for a behavioural intervention aimed at reducing cardiovascular risk.
Despite the efficiency offered by surrogate endpoints, there are concerns about their limitations, such as overestimating health benefits and possible failure to predict true benefits of interventions resulting in the roll out of interventions with no health benefit or even harmful. Therefore, design and reporting of RCTs using surrogate endpoints should clearly convey the uncertainty and risks associated with their use if critical. However, this has not been the case. For instance, an analysis of 626 RCTs found that 17% used a surrogate primary endpoint and of these, only 35% discussed whether the surrogate was a valid predictor of patient-relevant outcomes. Reporting guidelines can guide design and improve the reporting of RCTs at both the protocol and report stages. However, there is no consensus-driven guideline to report RCTs using surrogate primary endpoints. Our working definition of a surrogate endpoint is: ‘a biomarker or intermediate outcome used to substitute and predict for a final patient relevant outcome (i.e., characteristic or variable that captures how a patient feels, functions, or how long they survive, such as the outcomes of mortality or health-related quality of life)’.
Aims and phases of research
Therefore, this project aims to develop and disseminate CONSORT (Consolidated Standards of Reporting Trials) and SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) extensions to improve reporting of trials and protocols with surrogate primary endpoints.
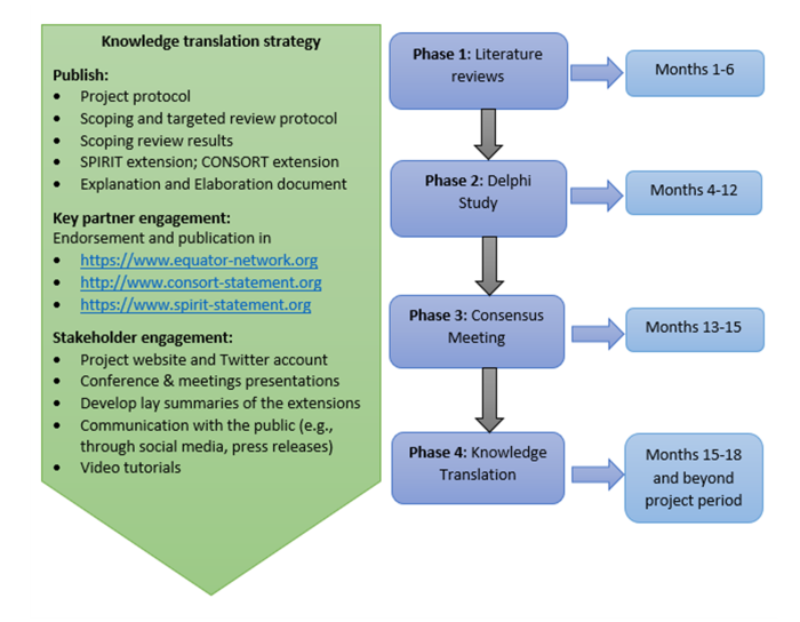
To achieve this aim, published guidance on development of health research reporting guidelines will be used. Based on this guidance, the project will have four phases:
- Phase 1 (Literature reviews) to identify items to be included in extensions and participants for a Delphi study;
- Phase 2 (Delphi study) to rate the items identified in Phase 1;
- Phase 3 (Consensus meeting) to ratify on rated items and agree on final items for inclusion in extensions;
- Phase 4 (Knowledge translation) to disseminate the project outputs through various strategies such as publications, key partners, and stakeholder engagement. The project activities will be conducted over 18 months starting from January 2022, (see Figure below).
Engage with us
Contact us: consort-surrogate@glasgow.ac.uk
Follow us on X @Consort_surr
Register your interest to participate in the project
Take part in the Delphi survey
Comments for each definition WORD
Comments for each rated item WORD
Cover letter summary and Round 1 feedback WORD
Endorsement & Funding
The project funding application was endorsed by Medical and products Regulatory Agency (MHRA) and the National Institute for Health and Care Excellence (NICE). The project is funded by the Medical Research Council (MRC) Better Methods for Better Health.
Project management group
- Dr Oriana Ciani, Principal Investigator, SDA Bocconi School of Management, Milan
- Professor Rod Taylor, Principal Investigator, School of Health and Wellbeing, University of Glasgow
- Dr Anthony Manyara, Project Manager, School of Health and Wellbeing, University of Glasgow
- Professor Jane Blazeby, Bristol NIHR Biomedical Research Centre, University Hospitals Bristol and Weston NHS Foundation Trust, and University of Bristol
- Dr Philippa Davies, Co-investigator, Population Health Sciences, Bristol Medical School, University of Bristol
- Professor Chris Weir, Co-investigator, Edinburgh Clinical Trials Unit, Usher Institute, University of Edinburgh
- Derek Stewart, Patient and Public Involvement Lead
Advisory Executive Committee members
- Professor Joseph Ross, Professor of Medicine (General Medicine) and of Public Health (Health Policy and Management) at the Yale School of Medicine; U.S. Outreach and Research Editor at the BMJ. (Chair of the Executive Committee)
- Professor Martin Offringa, Co-Chair of the international SPIRIT/CONSORT-Outcomes Group
- Dr. Nancy Butcher, Co-Chair of the international SPIRIT/CONSORT-Outcomes Group
- Professor An-Wen Chan, SPIRIT Group Representative
- Professor Gary Collins, EQUATOR (Enhancing the QUAlity and Transparency Of health Research) Network Representative
- Professor Sylwia Bujkiewicz, Academic statistician and surrogate endpoint evaluation expert
- Dr. Dalia Dawoud, Scientific Adviser at NICE
- Dr. Mario Ouwens, Senior Statistical Science Director and lead of the Statistical Innovation in Reimbursement Evidence and Real-World Evidence group within AstraZeneca; Chair of the International Society of Pharmacoeconomics and Outcomes Research taskforce on surrogacy.
Project updates
Endorsements and partner engagement
- Registration on the EQUATOR Network website on 10th March 2022 : https://www.equator-network.org/library/reporting-guidelines-under-development/reporting-guidelines-under-development-for-clinical-trials/#SURROGATE and https://www.equator-network.org/library/reporting-guidelines-under-development/reporting-guidelines-under-development-for-clinical-trials-protocols/#SURROGATE
Publications
- Commentary: Need for better reporting of trials with surrogate endpoints: SPIRIT|CONSORTSURROGATE extensions in the Journal of Epidemiology and Community Health, https://doi.org/10.1136/jech-2022-219294
- Commentary: Surrogate endpoints in trials: a call for better reporting https://doi.org/10.1186/s13063-022-06904-7
- Letter: A call for better reporting of trials using surrogate primary endpoints in journal of Alzheimer's & Dementia: Translational Research & Clinical Interventions, https://doi.org/10.1002/trc2.12340
- Reply: Reply to Commentary by Cummings (2022): Surrogate endpoints extend beyond biomarkers in the journal of Alzheimer's & Dementia: Translational Research & Clinical Interventions, https://doi.org/10.1002/trc2.12344
- Opinion: Surrogate endpoints in trials—a call for better reporting in the BMJ, https://doi.org/10.1136/bmj.o1912
- Editorial: Surrogate endpoints in cardio-thoracic trials: a call for better reporting and improved interpretation of trial findings in the European Journal of Cardio-Thoracic Surgery, https://doi.org/10.1093/ejcts/ezac449
- Protocol: Protocol for the development of SPIRIT and CONSORT extensions for randomised controlled trials with surrogate primary endpoints: SPIRIT-SURROGATE and CONSORT-SURROGATE in BMJ Open, https://doi.org/10.1136/bmjopen-2022-064304
- Protocol: Scoping and targeted reviews to support development of SPIRIT and CONSORT extensions for randomised controlled trials with surrogate primary endpoints: protocol, https://doi.org/10.1136/bmjopen-2022-062798

