Multi-Scale Models of Growth, Remodelling, and Disease in Soft Tissues
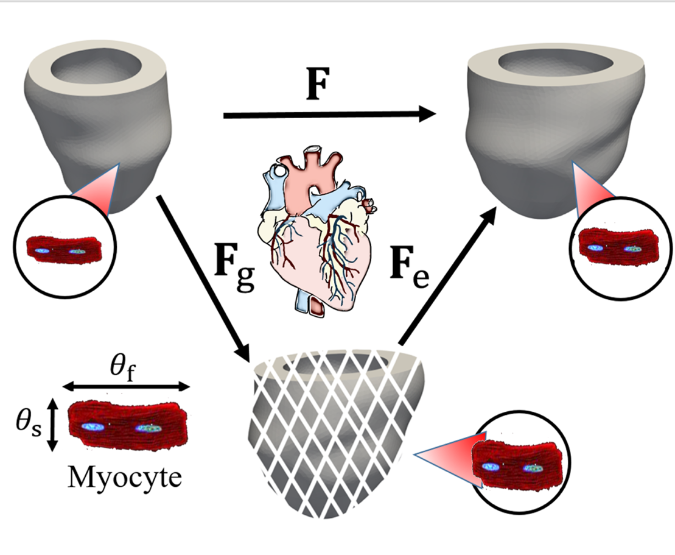
Maladaptive growth and remodelling in soft tissues are complex, multi-scale processes involving intricate physiological, neurohormonal, molecular, and cellular adaptations in response to internal or external environmental changes. Among these, mechanical stimuli have been considered as a key driver of disease progression in many soft tissue organs—for example, the heart. In the progression to heart dysfunction, mechanical overload, myocyte hypertrophy and fibrosis all trigger cascades of responses that impair cardiac function if persisted. We focus on myocardial adaptation during disease: how myocytes, fibroblasts, and other cardiac cell populations respond to biomechanical imbalance, and how those responses aggregate up to structural and functional adaption, and long-term adverse outcome. Multi-scale mathematical models are being developed by integrating cellular mechanics, constrained mixture theory, constituent-based constitutive modelling, and multi-physics approaches, aim to predict long-term outcomes of growth and remodelling under different disease scenarios (e.g. post-infarct healing, hypertrophy, fibrosis), to test interventions (surgical, pharmacological, mechanical), and to provide mechanistic insight into disease progression and possible therapies.
Researchers
Publications
- Constrained mixture models of growth and remodelling in an infarct left ventricle: A modelling study. Journal of the Mechanics and Physics of Solids, 200, (2025).
- Assessing left ventricular pump function using an immersed boundary method combined with finite elements. International Journal of Fluid Engineering, 2(2), (2025).
- Modelling the rheology of living cell cytoplasm: Poroviscoelasticity and fluid-to-solid transition. Biomechanics and Modeling in Mechanobiology, 23(5), (2024).
- An updated Lagrangian constrained mixture model of pathological cardiac growth and remodelling. Acta Biomaterialia, 166, (2023).
- Coupled agent‐based and hyperelastic modelling of the left ventricle post‐myocardial infarction. International journal for numerical methods in biomedical engineering, 35(1), (2019).

