VascuSens
Self-reporting vascular graft development.
Vascusens is a University of Glasgow research group developing innovative vascular sensing and therapy technology focused on implantable medical devices. This revolutionary technology will ultimately diagnose and treat clogged arteries before the blockage becomes symptomatic.
Developed in collaboration between Dr John Mercer (University of Glasgow) and Dr Steven Neale (University of Edinburgh), the platform technology functions by detecting both the quantity and quality of blockages that impact blood flow. The flexible design allows the sensor and telemetry to be integrated into a number of different iterations including stents and vascular grafts (figure 1). The sensor and implantable telemetry device collects impedimetric data from inside the body and relays it to the outside world for cloud based processing. The data is then formatted and processed to provide a visualization of inside the vessel. The clinically useful data is then relayed to an ios/android app developed by the group for both patient and clinician.
Cardiovascular disease will kill 1 in 3 patients suffering from heart attacks, strokes and other vascular complications caused by blocked arteries, leading to 17.5 million premature deaths globally per annum (WHO statistics 2020). Detecting these occlusions as they form inside the body is beyond current medical technology, making self-reporting implantable devices truly transformative for global healthcare. Initially, the group has developed a device for reporting vascular blockages (restenosis and thrombosis) found in chronic kidney disease (CKD) patients that require permanent vascular access. some 10% of the global population suffer from CKD and 2.5 million patients will require end stage renal dialysis. Although this figure accounts for 1% of affected patients, their cost of treatment is estimated to consume 8% of the medicare budget (an estimate of $100 billion). A self-reporting device that can conduct diagnostic surveillance would be transformational, reducing hospitalisation and treatment times and costs, but a device that can also offer a therapeutic element would be truly transformational for this cohort. Such a device would offer a personalised approach to treatment, reducing patient suffering and offering the optimal, most time-efficient treatment.
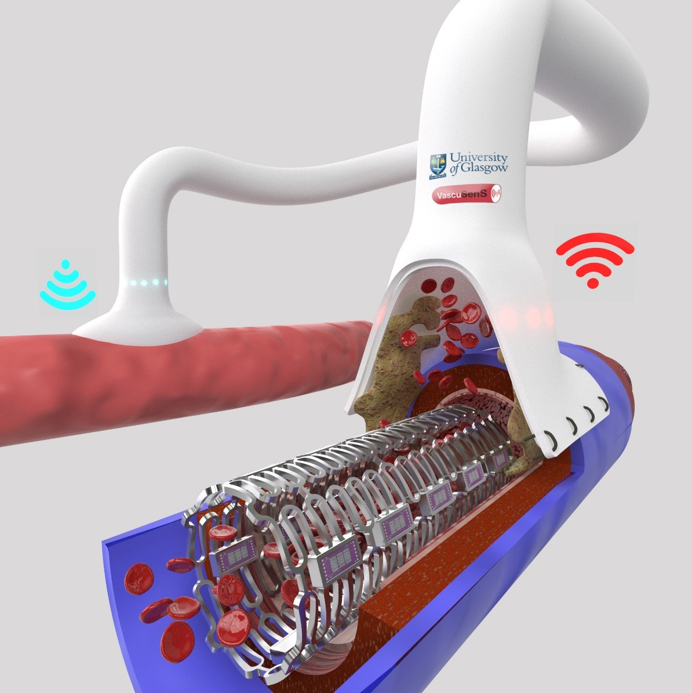
Figure 1. VascuSens; a non-invasive diagnostic and therapeutic technology integrated into implants that wirelessly report impending vascular complications in real-time.
The team behind VascuSens identified and worked with several commercial partners through sub-contracts that helped to refine and deliver this project. These partnerships allowed vascusens to scale up their product development, integrate their sensors into new vascular devices, and develop novel post processing techniques for their latest models (stage 3&4, concept progression & scale-up of product).
VascuSens has received funding from a wide variety of sources as well as in-kind support from companies and organisations, including the Wellcome Trust, the Medical Research Council (MRC), the British Heart Foundation (BHF) and the Chief Scientist Office (CSO) (funding opportunities). In 2020, the team signed a contract service for custom graft fabrication and regular supply of samples with Zeus Inc. Additionally, they developed contract services for custom miniaturised pcb and encapsulation processes. The team behind VascuSens have formed successful collaborations with industrial partners to assist with sensor encapsulation and assess the suitability for secondary processing of implants. Through their successful engagement with healthcare and commercial organisations, VascuSens have also secured a contract service for sensor array design and fabrication, and received clinical and expert regulatory advice along the way (stage 3, concept of progression). The group is currently completing preclinical trials and a full health economic assessment (FHEA) that is due to report about the economic viability of these devices in July 2022.
The college of MVLS provided additional support to this project by awarding VascuSens with the Reinvigorating Research Award (£40k). The University and Translational Research Initiative (TRI) team have supported VascuSens during the past few years, suggesting different funding avenues and assisting with their applications and successful securing of a variety of awards.
“Had it not been for the TRI and their support, we would not be at the stage that we are right now” – Dr John Mercer, lead investigator.
The VascuSens technology is currently patent pending. The team have also developed iOS/Android software for wireless interrogation and visualisation of data sets and have recently signed a new contract with a high-profile, multinational healthcare company to develop therapeutic aspects of the device. Currently, the team behind VascuSens are working hard to progress their work towards their future goal of integrating a therapeutic unit in their device as a single closed loop unit to both monitor and treat vascular pathologies.
The project was recognised by the Institute of Engineering and Technology (IET), gaining its lead PhD student Daniel Hoare the outstanding researcher of the year award and first prize of £10,000 for his work on our self-reporting cardiovascular devices. As part of this project, Daniel has also secured a Graham Wilson travel scholarship, and the Royal Society of Edinburgh Jim Lessells scholarship in 2020.
For more details about the ways the MVLS TRI can support your research, explore our different road maps on a variety of innovation themes here. For more information about VascuSens, contact Dr John Mercer. If you are seeking translational funding for your research, please contact Louise Mason and mvls-innovation@glasgow.ac.uk.
Partnership Funding
Starting from 2017, the research group received a BHF award, followed by CSO funding in 2018, and GKE and MRC CiC funding in 2019. The team were awarded a further MRC CiC award in 2020, that led to them securing an Industrial contract and Wellcome TPA funding in 2021. VascuSens has recently been supported by further awards, including the Reinvigorating research award, another MRC CiC and GKE, and have benefitted from TRI Device regulation champion support in 2022. Finally, they have secured a large contract with an industrial partner in 2022.
- 2017 - British Heart Foundation (BHF) £20K
- 2018 - Chief Scientific Office (CSO) £20K
- 2019 - Glasgow Knowledge Exchange Fund (GKE) £2K, MRC CIC Awards £85K
- 2020 - Continuing MRC CIC Award £38K
- 2021 - New Industrial Contract £55K, Wellcome TPA £10K
- 2022 - MRC CIC Follow on Award £24K, Glasgow Knowledge Exchange Fund (GKE) £30K, TRI Device Regulation Champion Support, Contract with Multinational Healthcare Company £90K


