Increasing vaccine access
Cattle vaccines against malignant catarrhal fever in East Africa.
Supported by the MVLS BBSRC Impact Acceleration Accounts (IAA) and by the Wellcome Trust Translational Partnership Award (TPA), Professor Sarah Cleaveland and collaborators are working to combine scientific research and policy development by studying the dynamics, impacts and implications of infections in natural ecosystems, with a focus on diseases in tropical countries.
Malignant Catarrhal Fever (MCF) is a fatal viral cattle disease that occurs following cross-species transmission from wildebeest calves (Figure 1). The implications of MCF are especially damaging to pastoral populations, as it leads to the loss of cattle, food, and a significant portion of their household income. With no currently available treatments, affected communities mainly rely on prevention measures. These include avoiding high-quality grazing lands during the wildebeest calving season at a time when this grazing is critical for cattle to conceive. Moving cattle away from their principal family residence not only affects cattle production and reproduction, but also reduces the availability of milk for the remaining family members (mainly women and children), who are heavily dependent on milk for energy and protein needs. Combined with the increased pressures on grazing lands as a result of climate change, the expansion of crop-based mechanised agriculture and the expansion of wildlife areas, MCF poses a threat to both local human and animal populations and the ecosystem.
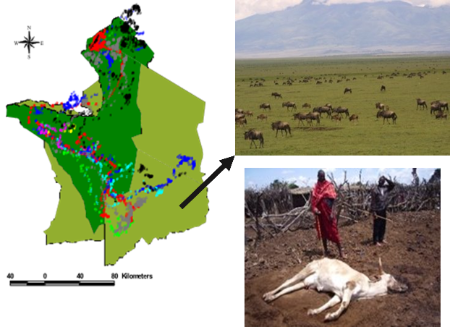
Figure 1. Map showing locations of wildebeest during the annual migration with calving occurring in mixed land-use areas (arrow) also utilised by pastoral livestock. Transmission of the MCF virus from wildebeest calves to cattle results in a fatal disease.
To solve this urgent unmet societal, environmental and economic need, Prof Cleaveland and the collaborative team have carried out a series of field studies in Tanzania on the safety and immunogenicity of a new attenuated cattle vaccine developed by the Moredun Research Institute in Scotland, the Nelson Mandela African Institution of Science and Technology, and Global Animal Health Tanzania, additional partners on the field research. This work follows earlier field trials in Tanzania that demonstrated a 56% protection against natural infection, and, in parallel studies in Kenya led by the International Livestock Research Institute, an 80% protection against fatal disease.
The studies showed high levels of vaccine safety and consistently high antibody responses in locally managed cattle regardless of body condition, sex, or reproductive status. Furthermore, antibody levels waned only slightly following vaccination, with all vaccinated animals showing a marked response to a single boost vaccination the following year. These results not only suggest excellent performance of the vaccine under field conditions, including a single dose boost vaccination. This allows for greater flexibility in the timing of vaccine delivery than originally anticipated and opens avenues for sustainable vaccine delivery which could provide significant benefits to affected communities.
A key element of these studies has been engagement with local communities, industry, and other stakeholders to explore their perceptions of the vaccine and willingness to adopt it, as well as to explore commercial opportunities for local production and larger-scale use of the vaccine in East Africa. The sustained interactions with communities and frequent feedback and discussion of results have built a strong platform of trust, with high levels of confidence in the vaccine, and enthusiasm for co-developing strategies for vaccine delivery (Figure 2).

Figure 2. Community engagement with local populations around vaccine adoption strategies.
Currently, the vaccine is covered by a licensing agreement between the Moredun Research Institute (MRI) and Onderstepoort Biological Products (OBP) for commercial production and use in South Africa. However, given the low likelihood that the vaccine will be fully registered in South Africa before 2026, the team are engaging in discussions with the Ministry of Livestock and Fisheries, local manufacturers and a translational product development organisation, GALVmed, to explore options for the use of the vaccine in East Africa, including under emergency license. Building on the results of the immunogenicity study, the team are also working to explore opportunities for co-delivery of MCF vaccine with other cattle vaccines routinely administered on an annual basis.
The significance of this project extends beyond the immediate benefits to MCF-affected communities in highlighting threats to pastoralist communities and the wide-ranging benefits of pastoralism in relation to food production, biodiversity and climate resilience. These benefits are increasingly being recognised by other agencies, including the Bill and Melinda Gates Foundation, but are at risk of being lost without concerted efforts to support and sustain these livestock production systems.

BBSRC Research Outcomes & Impact Case Study
Connecting climate change, livestock and zoonotic disease in Tanzania.
Funding Sources
This project has been supported by different internal awards. Early stages of the project were supported by BBSRC IAA, following by a Wellcome Trust Translational Partnership Award in 2021.




