Research
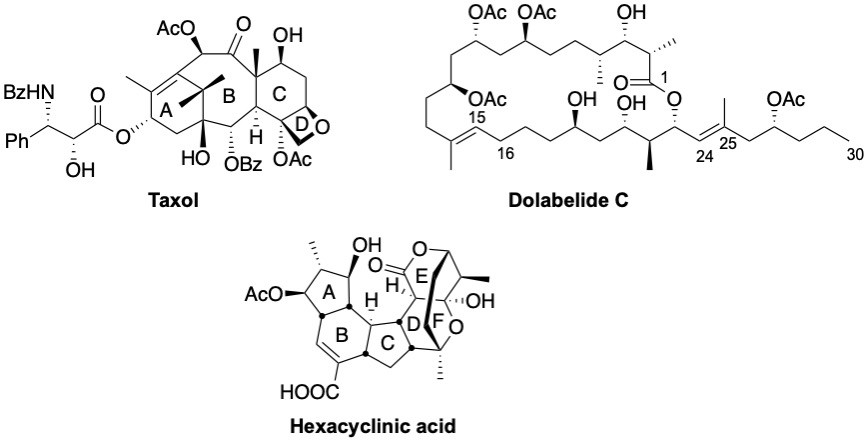
Our traditional research interest is the synthesis of biologically active natural products, such as taxol, a powerful antitumor agent, or dolabelide C and hexacyclinic acid, molecules with cytotoxic activity. In our approaches towards the target molecules, we uncover problems that cannot be easily solved by known reactions, and to overcome these difficulties we try to develop new methodologies that can also be applied to other syntheses.
Hexacyclinic acid
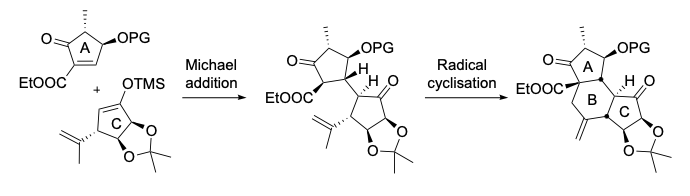
The only current project in this area is the synthesis of hexacyclinic acid. The ABC tricycle has been constructed using a highly diastereoselective Michael addition and a radical cyclisation as key steps.
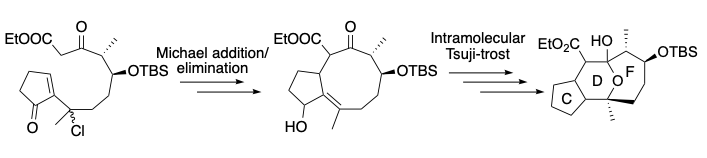
We have also achieved the synthesis of the CDF system. The key steps are a Michael addition/elimination sequence and an original intramolecular Tsuji-Trost reaction of an enol with an allylic alcohol.
Post-polymerisation functionalisation by olefin metathesis
The Prunet group has also been using metathesis reactions for the post-polymerisation functionalisation of a diverse array of polymers with pendent olefin groups.
In collaboration with Prof. Christophe Thomas (Chimie ParisTech), the CM of polyesters and diverse small molecule partners was used to afford functionalised polymers with a dramatic increase in their glass-transition temperature compared to the starting polyesters.
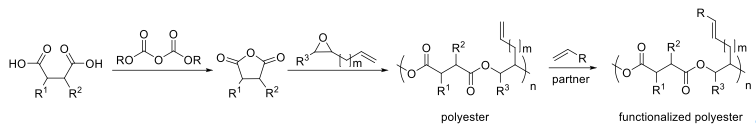
With Prof. Rob Liskamp (University of Glasgow), three novel polyethers with differently substituted olefinic side chains were prepared, tailored to promote cross metathesis (CM) vs self metathesis (SM), and the first successful CM between a polymer and a coupling partner of biological relevance, the tripeptide RGD, was performed.
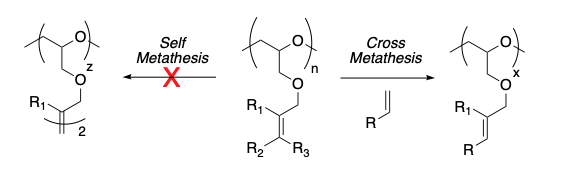
Insoluble beads of poly(divinylbenzene) prepared in the group of Prof. Peter Cormack (University of Strathclyde) could be functionalised by CM to provide ion-exchange resin materials.
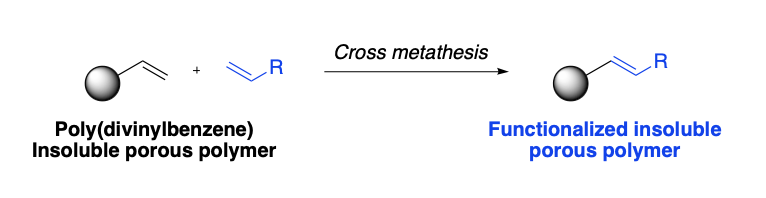
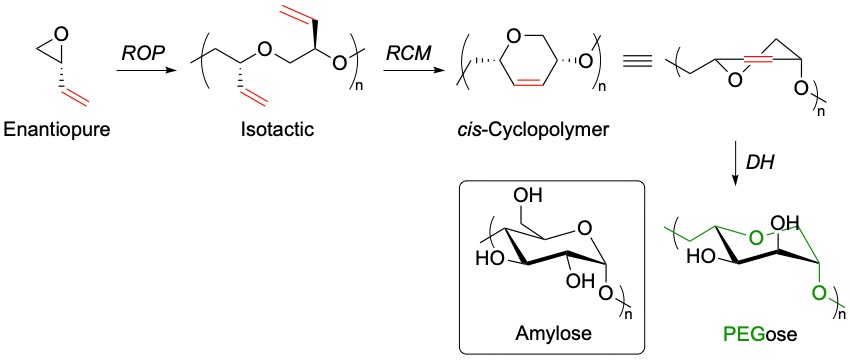
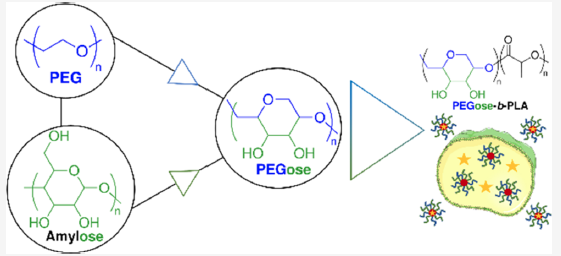
In collaboration with Prof. Michael Shaver (University of Manchester), RCM of isotactic polyepoxybutene (PEB) was used to prepare a stereocontrolled 1,4-linked cis cyclopolyether. Post-polymerisation dihydroxylation (DH) then furnished a poly(ethylene glycol) backbone with sugar-like functionalities (PEGose), which mimics the structure of amylose.
With Dr Bernhard Schmidt (University of Glasgow), we explored the use of PEGose as a PEG alternative. Nanoparticles composed of PEGose and poly(lactic acid) (PLA) were formed with narrow dispersity (150-200 nM). They were able to encapsulate and slowly release hydrophilic (Rhodamine) and hydrophilic (Nile Red) dyes. These nanoparticles were very stable, and exhibited low toxicity and a high propensity to penetrate cells, making them good candidates for drug delivery.
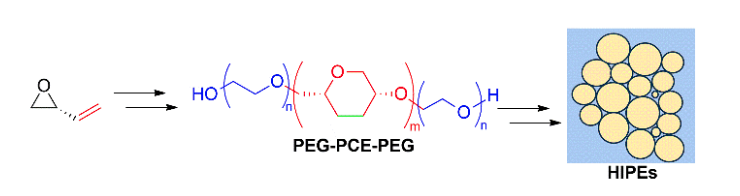
The polycycloether precursor of PEGose, after hydrogenation, was also used as an alternative of poly(propylene glycol) for the synthesis of poloxamer mimics with a rigid helical block. The formation of high internal phase emulsions (HIPEs) was observed with these triblock copolymers, irrespective of the oil/water ratio, which is not the case with the commonly used poloxamers P188 or P407.