New research could help lithium-ion batteries reach their full potential
Published: 5 March 2026
New research into why lithium-ion batteries tend to lose power over cycles of charge and discharge could help unlock the next generation of more durable, fast-charging power cells.
New research into why lithium-ion batteries tend to lose power over cycles of charge and discharge could help unlock the next generation of more durable, fast-charging power cells.
An international team of chemists and engineers from the UK, China, and Germany have published a paper which challenges the conventional assumptions about how batteries should be optimised to maximise performance, highlighting the importance of electrical, rather than ionic, conductivity.
As lithium-ion batteries become more widely adopted across industries from mobile computing to electric cars, researchers have been working to improve their long-term performance and their ability to charge quickly.
In the past, a key focus for research has been on battery’s ionic conductivity, which governs the speed that lithium ions move through the battery’s electrodes. The consensus assumption has been that fast-travelling ions lead to better-performing batteries.
Relatively little attention has been paid to the role of another property of lithium-ion battery cathodes in determining their longevity: their electronic conductivity. In a paper published in early view in the journal Advanced Science, the University of Glasgow-led research team describe how they harnessed the unusual properties of chromium diselenide to isolate and study that property in-depth.
Their results suggest that electronic conductivity plays a crucial role in determining how much power a battery can deliver and how it behaves during fast charging. The finding could help lithium-ion batteries reach their full potential in the years to come.
Dr Alexey Ganin, a senior lecturer in the University of Glasgow’s School of Chemistry, is the paper’s corresponding author. He said: “It’s often assumed that the main limit to the power of lithium-ion batteries is their ionic conductivity, or how fast lithium moves through the material the electrode is made from. What we’ve shown here is that the material’s electronic conductivity, which governs how easily electrons move, is just as important, if not more so.
“What makes this study powerful is the way the computational and experimental data agree. We predicted the conductivity changes first, then confirmed them at every scale, from muons to X‑rays to battery cells.”
The team chose chromium diselenide for their experiment because its unusual structure is unaffected by cycles of lithium ions moving through them. In many battery materials, the structure can change over time, making it difficult to isolate the effect of a single variable.
At the same time, lithium ions move through chromium diselenide extremely quickly, as fast as in some of the best battery materials used today. That made it an ideal candidate to probe how electronic conductivity alone might affect a battery’s performance over time.
Using computer modelling and a battery of experimental techniques carried out at facilities including the ISIS Neutron and Muon Source and Diamond Light Source at STFC’s Rutherford Appleton Laboratory in Oxfordshire, the researchers tracked in real time what happens inside the material as a battery operates.
The results showed that the material’s lithium conductivity remains high throughout repeated charging and discharging, confirming that the ionic conductivity is not the source of battery’s declining performance. Instead, the evidence points strongly to electronic conductivity.
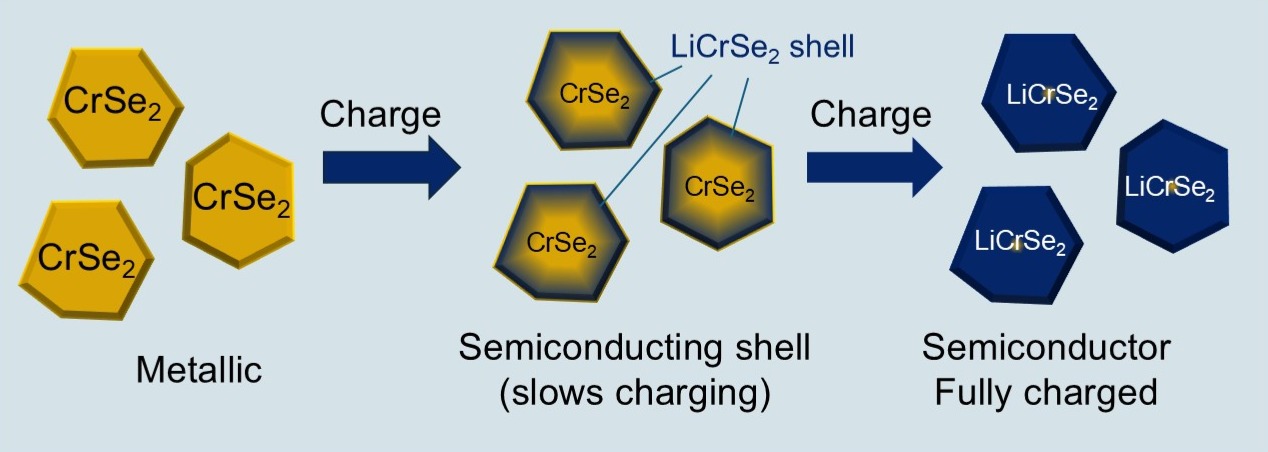
As the battery charges, lithium gradually works its way into the chromium diselenide particles from the outside in. The outer shell of each particle becomes fully lithiated and steadily less able to conduct electrons as it becomes electrically semiconducting.
For a short time, the interior, unreacted core remains metallic and highly conductive, preserving overall performance. But as lithiation progresses and the insulating shell thickens, a threshold is crossed: the electronic conductivity of the whole particle drops sharply, and with it, the voltage the battery can sustain. That shift is driven by the conductivity transition, the team suggests.
Dr Ganin added: “While our results won’t lead to chromium diselenide being used as cathodes in commercially-available batteries in the future, it’s an important insight into how we might overcome one of the key barriers to improving the performance of lithium-ion batteries.
“Electronic conductivity is something that can be calculated computationally for almost any material, at any stage of lithiation, without the need for expensive experiments. Our take-home message from this research is simply this: measure the electronic conductivity of your material and try to improve it, and we will quickly start to gain more insight into how batteries can be improved.”
The research was carried out in collaboration with theoretical chemists at Ulm University in Germany, who performed the computational work, as well as experimental groups at Queen Mary University of London, the University of Kent, and Tianjin University of Technology in China.
The team’s paper, titled ‘Lithiation-Driven LiCrSe2 Shell Growth on Metallic CrSe2 Core Governs the Plateau–Slope Behavior’, is published in Advanced Science.
The research was supported by funding from UKRI’s Engineering and Physical Sciences Research Council (EPSRC) and the DFG, German Research Foundation.
First published: 5 March 2026

