Dr Catherine Berry
- Reader (Molecular Biosciences)
telephone:
01413308400
email:
Catherine.Berry@glasgow.ac.uk
Room B3-09, Centre for Cell Engineering, IMCSB, Joseph Black Building, University Avenue, University of Glasgow, Glasgow, G12 8QQ
Biography
Catherine is a lecturer in the Centre for Cell Engineering in the University of Glasgow. Following her PhD at Queen Mary, University of London, was focused on tissue and bioengineering, she started in Glasgow in 2002, focusing on the use of inorganic nanoparticles for cell imaging and as delivery vehicles in cell culture models.
A range of her current projects include (i) the use of an external magnetic field to increase cell loading with magnetic nanoparticles for higher transfection efficacies, both in monolayer and 3-D cell culture models; (ii) manipulation of magnetic particles loaded MSCs & HSCs in 3-D culture to recreate a niche model system; (iii) a 3-D model for magnetic hyperthermia testing for cancer treatment; (iv) the knockdown of cell proliferation genes in cancer cells via gold nanoparticle mediated siNRA delivery; (v) the manipulation of mesenchymal stem cell differentiation via gold nanoparticle mediated inhibitors of crucial microRNAs.
Research interests
A strong and promising role for nanoparticles is foreseen in future biomedicine. Catherine’s current projects focus on using either magnetic NPs or gold NPs for applications in biomedicine. Both types of NP are regarded as safe for use in vivo, and benefit from their ease of synthesis and functionalisation, in addition to conferring useful optical and physical properties.
Current magnetic NP projects include:
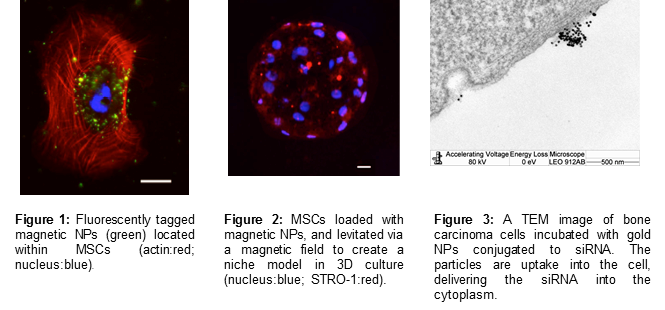
(i) The use of an external magnetic field to increase cell loading with magnetic nanoparticles for higher transfection efficacies, both in monolayer and 3-D cell culture models (figure 1)
(ii) Manipulation of magnetic mesenchymal stem cells (MSCs; pre-loaded with magnetic nanoparticles) into spheroid cultures, cultured within a collagen gel, to generate a quiescent, physiologically relevant model of the bone marrow niche (figure 2). This project has been extended to incorporate hematopoietic stem cells (HSCs) in the niche model system, co-cultured with osteoblasts or endothelial cells, to recreate the vascular and endosteal bone marrow niche environments respectively.
(iii) Adapting the bone marrow model developed in (ii) to investigate the invasion and remodelling of the bone marrow niche by leukaemic stem cells (LSCs), using magnetic 3D MSC/HSC and LSC co-cultures.
(iv) Adapting the quiescent bone marrow model developed in (ii) to design a dormant and recurrent 3D breast cancer cell model via magnetic spheroid co-culture of MSCs and breast cancer cells.
Current gold NP projects focus on the delivery of small molecules to cells and testing of photothermal therapy in cancer cells:
(i) The knockdown of cell proliferation genes in cancer cells via gold NP mediated siNRA delivery (figure 3).
(ii) The manipulation of MSC differentiation, e.g. enhancing osteogenesis, via gold NP mediated delivery of crucial microRNAs and/or their inhibitors (miRs/antagomiRs). This project has been extended to identify whether such NP/miR treatments can be used with osteoporotic MSCs.
(iii) Inducing cell death (2D and 3D culture) via laser heating of gold NP (nanoprisms and nanrods) loaded cancer cells.
Research groups
- Regenerative Medicine & Cellular Microenvironment
Grants
Grants and Awards listed are those received whilst working with the University of Glasgow.
- MAINSTREAM: EPSRC Research and Partnership Hub for Health Technologies in Manufacturing Stem Cells
Engineering and Physical Sciences Research Council
2025 - 2031
- Engineering the bone marrow niche to control stem cell regulation, metastaticevolution and cancer dormancy
Engineering and Physical Sciences Research Council
2023 - 2028
- Mechanobiology-based medicine - Phase 2
Engineering and Physical Sciences Research Council
2023 - 2028
- Engineered haematopoietic stem cell niches for disease modelling
National Centre for the Replacement of Animals Research
2020 - 2020
- Nanoparticle-Mediated microRNA Delivery for Increased Osteogenesis in a 3D Osteoporotic Bone Marrow Model.
Chief Scientist Office (CSO)
2018 - 2020
- Investigation of strategies to enable objective quantification of neural regeneration through tissue-engineered conduits in vivo. Preparing for clinical trials.
Royal College of Surgeons of Edinburgh
2016 - 2017
- Nanoparticles and nanotopography: a nano-toolbox to control stem cell self-renewal via microRNAs
Biotechnology and Biological Sciences Research Council
2014 - 2016
- Molecular interactions of Mannheimia haemolytica with the bovine and ovine respiratory tracts using three-dimensional tissue engineering approaches
Biotechnology and Biological Sciences Research Council
2014 - 2018
- Development of a three-dimensional air-liquid interface epithelial cell model to study pathogen interactions within the bovine respiratory tract
National Centre for the Replacement of Animals Research
2014 - 2017
- Characterisation of Mesenchymal Stem Cells in a 3D Niche Mimic via Microarray (ISSF)
Wellcome Trust
2013 - 2013
- IAA-EPSRC: Magnetic protein mediated hyperthermia in 3D cell culture
Engineering and Physical Sciences Research Council
2012 - 2015
- Nanoparticle Invasion in a Three-dimensional Human Skin Tissue Model.
The Royal Society
2010 - 2011
- Functionalised Gold and Semiconductor (quantum dot) nanoparticles for applications in cell and tissue engineering
The Royal Society
2009 - 2012
- Multifunctional gold nanoparticles for gene therapy
Engineering and Physical Sciences Research Council
2009 - 2012
- Determination of fluorescent quantum dot uptake by human cells in culture
Tenovus Scotland
2009 - 2010
- NANOSAFE 2 - safe production and use of nanomaterials
European Commission
2005 - 2009
Supervision
Catherine is interested in carrying out include developing new 3D multiwell plate culture platforms for HSCs, in order to support growth and retain primitive phenotype. She aims to do this in several ways, including collaborating with Prof John Christie to use optogenetics to control the expression of key co-cultured MSC phenotype markers in addition to key cytokines involved in HSC support and retention within the bone marrow. Such research involves the use of magnetically labeled MSCs to generate quiescent spheroid cultures, which can subsequently be manipulated via blue light to enhance MSC phenotype and/or cytokine release, to generate HSC-permissive environments.
In addition, she wishes to use the recently developed MSC/HSC multiwell plate cultures as high throughput platforms for assessing small molecules and drugs, for example looking at leukaemia treatments with LSC co-cultures.
Further developments using gold NPs for photothermal therapy (cancer treatments) will also be carried out, whereby specific temperature profiles can be used to more accurately apply laser technology to irradiate NP labeled cells.
- Duncan, Elaine
Bioengineering 3D Adipose Organoids for Type 2 Diabetes Drug Discovery - Jackson, Emma
Magnetic Hydrogels for Bone Tissue Engineering - Lee, Rui Ling
Bioactive 3D Hydrogel-based Bone Marrow Niche Model to Study Extracellular Vesicle-mediated Cell Communication in Acute Myeloid Leukaemia
Several projects have recently been completed, either evidenced as published articles or manuscripts in preparation.
With regard to magnetic NPs, completed projects include the characterisation of MSC spheroids in 3D culture (Lewis NS et al, J Tiss Eng, 2017) and the confirmation that MSC spheroids in 3D can respond appropriately to co-culture cell injury via appropriate migration and differentiation (Lewis EEL et al, ACS Nano, 2016).
With regard to gold NPs, we have successfully studied the gold NP-mediated delivery of siRNA for c-myc knockdown in cancer cells (Child HW et al, Nanomedicine, 2015; Conde J et al, ACS Nano, 2015) in addition to optimizing the gluoathione/polyethylene glycol ratios for successful siRNA delivery and release from gold NPs within cells (McCully M et al, Nano Research, 2015).
Teaching
Catherine is involved in teaching to both undergraduate and postgraduates at Glasgow. Her main UG teaching includes running a level 3 lab (genetics & MCB students) and lecturing on the level 4 Cell Engineering and Stem Cell options (deputy course coordinator for both options). With regard to PG teaching, she is involved in translations skills tutorials to a cohort of PhD students and teaching on Stem Cell summer school courses. She currently primarily supervises four PhD students, and co-supervises three additional PhD students, as well as regularly supervising Masters and UG students.
Additional information
Editorial Board
- 2015 - present: Frontiers in Bioengineering & Biotechnology - editorial board member
- 2007 - 2008: IEEE Transactions in Nanobioscience - Guest Editor
- 2005 - present: Current Nanoscience - Board member
Grant Advisory Board
- 2016 - present: Medical Research Scotland - PhD Studentship Panel
Invited International Presentations
- 2009: Glasgow, Scotland - Keynote Speaker - Nexxus Scotland Flexible Working
- 2009: Glasgow, Scotland - Keynote Speaker - Tissue & Cell Engineering Society
- 2007: Nantes, France - European Society Biomaterials
- 2005: London - Keynote Speaker - UK Magnetics Workshop
- 2004: London - Keynote Speaker - EPSRC Nanomagnetics Workshop
- 2004: London - Materials Congress
- 2003: Cardiff - Tissue & Cell Engineering Society
- 2003: Invited speaker Particles Interface Group
- 2002: Leeds, UK - UK Society Biomaterials
- 2002: Glasgow, Scotland - Co-organiser and co-chair: Tissue and Cell Engineering Society Meeting
- 2002: Barcelona, Spain - European Society Biomaterials
- 2002: Glasgow, Scotland - Tissue & Cell Engineering Society
Research Fellowship
- 2006 - 2010: Royal Society Dorothy Hodgkin Fellowship



