Iron overload can actively contribute to ongoing brain damage in PMS
Published: 31 May 2022
Research by Institute of Infection, Immunity and Inflammation scientists, led by Dr Katja Thümmler, has shown for the first time that iron overload can actively contribute to the ongoing brain damage in progressive multiple sclerosis (PMS), confirming that treatments designed to prevent this iron-related injury could slow accumulation of disability in PMS patients.
Research by Institute of Infection, Immunity and Inflammation scientists, led by Dr Katja Thümmler, has shown for the first time that iron overload can actively contribute to the ongoing brain damage in progressive multiple sclerosis (PMS), confirming that treatments designed to prevent this iron-related injury could slow accumulation of disability in PMS patients.
Multiple sclerosis (MS) as a major cause of chronic disability amongst young adults in Scotland can be devastating for patients, potentially resulting in having to end employment early and becoming wheelchair bound.
Although current treatments can help reduce some symptoms, they fail to halt accumulation of disability in patients with progressive MS, a form of the disease when nerve cell axons - the brain’s electrical wires - are permanently damaged.
This reflects an inadequate understanding of the mechanisms responsible for injury to axons in MS. It has, however, become clear that this injury can be caused by chronic oxidative stress, manifesting in the production of ‘free radicals’ that are highly toxic to cells in the brain.
Recent studies now demonstrate that this is due to an increased accumulation of iron in the brain of patients with PMS.
Iron is generally considered good – it is required, for instance, in our red blood cells to carry oxygen around the body and if we have abnormally low levels of iron we feel weak and tire easily. However when iron accumulates in the brain, as it does in PMS, it can become harmful.
Although a pathological role of iron overload in some neurological diseases has been suggested, it is rarely considered in the context of MS.
In this research project, published in Experimental Neurology and funded by Multiple Sclerosis Society UK and TENOVUS Scotland, iii scientists determined exactly how iron injures the brain’s electrical wires and how to stop its damaging effects.
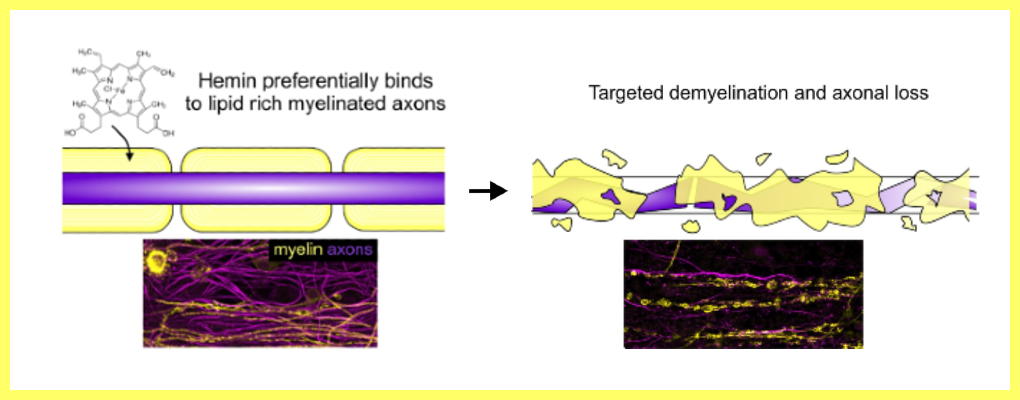
As suggested by studies indicating that some MS patients have an increased frequency of 'microbleeds' in the brain, they propose that brain iron can be derived from breakdown of haemoglobin, the substance in red blood cells that packages iron.
By using a 'brain-in-a-dish' model of MS developed in Glasgow, they have now, for the first time, shown that increased iron or iron-containing haemoglobin breakdown products indeed selectively damage nerve cell axons and their fatty insulation (myelin), whilst sparing other cells of the central nervous system.
This new concept of a targeted effect of iron was quite a surprise, since it was believed that iron is more or less equally toxic to all cell types.
Dr Thümmler said: “Seeing this targeted detrimental effect of iron on the same structures injured during the course of MS was really exciting, because we immediately knew that iron overload is not just a bystander effect of the general ongoing brain damage but iron does in fact actively contribute to the pathology of MS.”
Interestingly, the scientists also found that iron bound to haemoglobin breakdown products is much more toxic to brain cells than free iron.
In a big screening of several potential mediators of this detrimental iron effect, they discovered that iron overload induces a specific type of iron-regulated cell death (ferroptosis) and an increase in damaging reactive oxygen species plays an important role.
This concept is important as it tells us that treatments that prevent this iron-related injury could be able slow accumulation of disability in PMS, but might also play an important role in other neurological diseases in which iron/haemoglobin breakdown products are implicated such as Alzheimer’s disease, Parkinson’s disease and most importantly haemorrhagic stroke.
- Karl Baldacchino, William J.Peveler, Leandro Lemgruber, Rebecca Sherrard Smith, Cornelia Scharler, Lorna Hayden, Lina Komarek, Susan L. Lindsay, Susan C. Barnett, Julia M.Edgar, Christopher Linington, Katja Thümmler
- First Published: 13 May 2022. Experimental Neurology, Volume 354, August 2022, 114113.
- DOI: https://doi.org/10.1016/j.expneurol.2022.114113.
Image Legend: Graphical abstract
Funding: Multiple Sclerosis Society UK and TENOVUS Scotland
First published: 31 May 2022

