Deadly parasites create unique cellular structures to survive
Published: 7 January 2021
Scientists have solved a key parasitic puzzle, revealing the unique and complex structures toxoplasmosis and malaria parasites make in order to survive in different hosts.
Scientists have solved a key parasitic puzzle, revealing the unique and complex structures toxoplasmosis and malaria parasites make in order to survive in different hosts.
The new study, led by the University of Glasgow in collaboration with the University of Stockholm, and published in Nature Communications, details how certain parasites can create unique cellular structures to control how they create energy and thus survive in different hosts.
Malaria and toxoplasmosis, both potentially deadly diseases, are caused by similar parasites which organise themselves to exploit their host’s energy resources in order to infect and transmit to new hosts. However, until now, scientists didn’t fully understand the detailed mechanisms behind this process.
In this new research, researchers have solved a parasitic puzzle at the heart of how these deadly pathogens are able to survive in different hosts in order for them to transmit onwards.
Toxoplasmosis is a disease caused by the Toxoplasma parasite, and thought to be carried by an estimate 33% of the global population in its dormant state. However, in those with weakened immune systems this parasite can ‘wake up’ and cause complications such as stroke and brain damage. Malaria, a mosquito-borne infectious disease, currently affects over 200 million people, and kills nearly half a million people – mostly children – every year.
In order to survive these parasites rely on resources available in their host – for toxoplasmosis it is animals and humans, while for malaria this includes also insects. This means that in order to survive, to infect the host and to transmit between hosts, these parasites have to be flexible in how they create energy based on what is available to them.
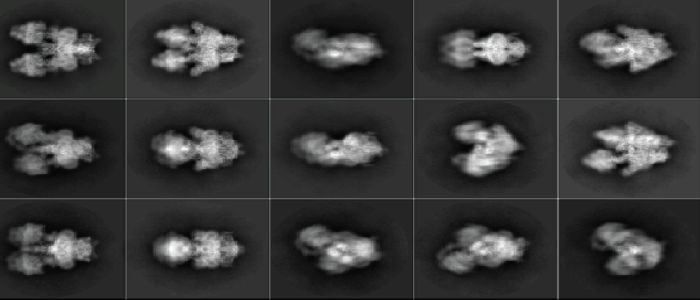
Scientists studied a vital energy-producing machine within the parasite called ATPsynthase. In addition to making energy, ATPsynthase machines can also come together into large structures that together shape the mitochondrial membrane, controlling the rate of energy production, and key to its survival. Researchers found that, in these parasites, the ATPsynthase machines were able to make complex and unique pentagonal pyramid structures, unlike anything produced by the same systems in their human host.
Dr Lilach Sheiner, on the lead authors of the study from the University of Glasgow, said: “We have made major progress in understanding how the parasites which cause toxoplasmosis and malaria can adapt the way they make energy to the environment they experience in their host. This is critical for the parasite’s ability to disseminate in different tissues and to transmit between hosts.
“In addition to understanding better how these parasites manage to survive and maintain flexibility while navigating different host environments, these findings have importance in terms of generating knowledge that can inform drug discovery. Our structure can now be used for "digitally" screening for parasite-specific inhibitors – a promising strategy because it is so different to the human structure.”
Toxoplasmosis is generally transmitted through undercooked meat, soil, or from contact with cat faeces. Although it is estimated that 33% of the population in the UK carry a dormant form of the parasite, symptoms of infection in healthy adults generally go unnoticed.
However, toxoplasmosis can be dangerous to unborn children and in people with compromised immune systems, such as patients with AIDS. When the Toxoplasma ‘wakes up’ in people with compromised immune systems it can cause stroke, and in infants it can cause severe brain damage.
Malaria is caused by the related parasite, Plasmodium, which infects humans through the bite of a mosquito. The parasite then grows in the liver and in red blood cells in our blood. Parasites can also change in the blood to take on a male and female form, which can re-infect mosquitoes when they bite and suck blood from infected people.
The paper, ‘ATP synthase hexamer assemblies shape cristae of Toxoplasma mitochondria’ is published in Nature Communications. The work was funded by Swedish Foundation for Strategic Research, Ragnar Söderberg Foundation, Swedish Research Council, Cancerfonden, European Research Council, Knut and Alice Wallenberg Foundation, The BBSRC, and A Wellcome Investigator award (217173/Z/19/Z). Dr Lilach Sheiner is a Royal Society of Edinburgh Personal Research Fellow.
Enquiries: ali.howard@glasgow.ac.uk or elizabeth.mcmeekin@glasgow.ac.uk / 0141 330 6557 or 0141 330 4831
First published: 7 January 2021

