Research interests
G PROTEIN COUPLED RECEPTORS
We were among the first laboratories to determine that GPCRs undergo rapid phosphorylation following agonist addition. Primarily using the five members of the muscarinic receptor family as an exemplar GPCR class we have successfully applied phospho-proteomics, phospho-specific antibodies, receptor mutagenesis and cell signalling to define the signalling role of receptor phosphorylation. In these studies we described the phosphorylation bar-code that contributes to the signalling out-put of many GPCRs.
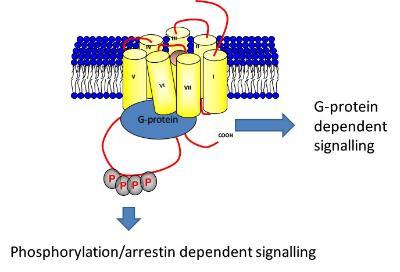
Biased Signalling: We have recently extended our work on GPCR phosphorylation to investigate the in vivo role of GPCR phosphorylation. By mutating the phosphorylation sites on the M3-muscarinic acetylcholine receptor (M3-receptor) gene we have generated the first genetic model of a G protein biased receptor – a receptor that is coupled to heterotrimeric G proteins but uncoupled from receptor phosphorylation/arrestin signalling. In a series of papers published in PNAS we have defined the physiological responses of the M3-receptor that are mediated either by receptor phosphorylation or G protein signalling. In so doing we have generated the first in vivo signalling map of a GPCR thereby providing a model to predict the effect of biased GPCR ligands.
Neurodegeneration: To establish the validity of the M1-receptor as a target in neurodegeneration we have analysed mouse prion disease for features that map onto those observed in human Alzheimer’s disease (AD). In these studies we have established that mouse prion disease is a progressive terminal disease that is associated with progressive neuronal loss, particularly of cholinergic neurones of the hippocampus, which results in memory loss that can be reversed with clinical acetylcholine esterase inhibitors - as is the case in AD. Furthermore, late stage prion disease hippocampus expresses normal levels of muscarinic receptors that retain normal signalling and sensitivity to muscarinic drugs. Using this model we have been able to show the following:
• That muscarinic allosteric and orthosteric ligands can completely rescue the memory deficit in prion diseased mice
• That prolonged administration of M1-receptor allosteric modulators can slow the progression of terminal prion disease.
We are currently employing genetic and chemical genetic approaches as well as novel pharmacological ligands, advanced imaging and protein and RNA ‘omic approaches to study the molecular mechanisms of the action of muscarinic ligands in memory loss in neurodegeneration and to determine the mechanism of muscarinic-mediated prion disease modification.
Free fatty acid receptor (FFA) family: In collaboration with Professor Graeme Milligan we have BBSRC funded programmes to investigate the physiological and patho-physiological role of the FFA family of GPCRs. This receptor family consists of 4 members FFA1-FFA4. In this family FFA1 and FFA4 respond to long chain fatty acids such as the essential omega-3 fatty acids found in fish oils, and the FFA2 and FFA3 receptors respond to short chain fatty acids such as acetate, butyrate and proprionate that are generated by the gut microbiota. Using novel pharmacological ligands in concert with genetic animal models we have defined new roles for this receptor family. In particular, we have established that the FFA4 receptor expressed in airway epithelium mediates airway smooth muscle relaxation. We are currently engaged in a programme investigating the validity of FFA4 as a novel target in human inflammatory airway diseases such as asthma and chronic obstructive pulmonary disease (COPD).
Neglected and hard to target GPCRs: In a programme of research closely associated with the recently established at Glasgow we are collaborating with industrial partners and academic laboratories to establish the signalling properties, in vivo function and therapeutic potential of hard to target GPCRs. These include receptors such as GPR35 and GPR84.
MALARIA RESEARCH
Applying much of the pharmacological, cell biological, genetic and proteomic approaches developed in the study of mammalian cell signalling the we are investigating essential phospho-signalling cascades in malaria as well as contributing to a world-wide network to define mechanisms of drug resistance in malaria.
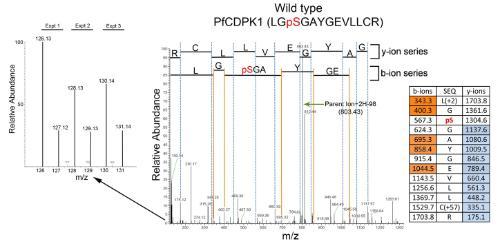
Essential malaria parasite protein kinases: Working together with Professor Christian Doerig we were among the first laboratories to publish the global phospho-proteome of the most lethal strain of human malaria parasite, P. falciparum. We also determined the protein kinases that were essential for the survival of blood stage P. falciparum. These studies formed the basis of our subsequent drug discovery programmes targeting protein kinases that we know are important to maintain the parasite.
Targeting the PfCLK-family: In collaboration with GSK we have an active programme to determine the fundamental biology and therapeutic potential of targeting the four members of the PfCLK family. Considered to be important in RNA processing we have screened GSK (TCAMS) molecules that are known to have anti-malarial activity as inhibitors of the PfCLK family. These studies have defined unique inhibitors that kill the parasite at various life stages through inhibition of PfCLK3. In this way we have not only validated PfCLK3 as a novel target in malaria but also determined novel chemical inhibitors that could act as start points for drug development.
Chemical genetic approaches: We have been developing chemical genetic approaches to probing the function and potential therapeutic value of targeting protein kinases in malaria. This has involved screening for inhibitors to particular protein kinases and then establishing mutations that render the kinase insensitive to the inhibitor. Generating a genetically modified parasite strain that expresses the mutant kinase in place of the wild type kinase provides a method of not only determining if inhibitors kill the parasite by targeting the protein kinase but also will establish the on and off target activity of the inhibitors allowing us to use the inhibitors as probes for kinase function.
Malaria resistance: We have joined a worldwide consortium of scientists investigating the mechanisms of artemisinin resistance associated with mutations in the K13-kelch protein. We have been employing mass spectrometry based global proteomics to establish the changes in the proteome associated with parasite resistance.
COLLABORATORS
We collaborate widely with the academic and industrial community. The list below are collaborators with whom we have obtained funding. In addition, we have many interactions that are not underpinned by specific funding schemes but nevertheless have resulted in highly productive long term research.
Academic:
• Professor Graeme Milligan, University of Glasgow. BBSRC and MRC funding.
• Dr Jonathon Taylor, University of Glasgow. PhD studentship.
• Professors Christopoulos and Sexton, Monash University: Wellcome Trust Collaborative Award.
• Dr Davis Nwakanma, MRC Unit the Gambia. PhD studentship.
Industrial:
• AstraZeneca. BBSRC Industrial Partnership Award and Open Innovation Agreement.
• Heptares Inc. ORBIT award.
• GlaxoSmithKline. Open Lab Foundation.
• Eli Lilly. MRC Industrial Collaboration Award.
PEOPLE
Lord Kelvin Adam Smith Fellows:
Sophie Bradley
Mahmood Alam
Post doctoral scientists:
Karen Thompson – Wellcome Trust
Louis Dwomoh – Wellcome Trust
Paulina Valušková – Wellcome Trust
Joe Lin -BBSRC
Tez Quon-BBSRC
Rudi Prihandoko-BBSRC
Ana Sanchez -Internal
PhD Students:
Omar Janha – MRC
Simon Brooke – ITTP
Research support:
Colin Molloy
Selected Publications:
1. Bradley SJ, Bourgognon JM Sanger HE Verity N, Mogg AJ, White DJ, Butcher AJ Moreno JA, Molloy C, Macedo-Hatch T, Edwards JM, Wess J, Pawlak R, Read DJ, Sexton PM, Broad LM, Steinert RJ, Mallucci GR, Christopoulos A, Felder CC, Tobin AB. M1-muscarinic allosteric modulators slow prion neurodegeneration and restore memory loss. J. Clin. Invest. (2017) 127(2):487-499.
2. Mitcheson DF, Bottrill AR, Bayliss R, Carr K, Coxon CR¶, Cano C, Golding BT¶, Griffin RJ, Fry AM, Doerig C, Tobin AB. A new tool for the chemical genetic investigation of Pfnek2 in Plasmodium falciparum. Malaria Journal. (2016) 15(1):535.
3. Prihandoko R, Alvarez-Curto E, Hudson BD, Butcher AJ, Ulven T, Miller AM, Tobin AB, Milligan G: Distinct phosphorylation clusters determines the signalling outcome of the free fatty acid receptor FFA4/GPR120. Mol Pharmacol (2016) 89(5):505-20.
4. Nuber S, Zabel U, Lorenz K, Nuber A, Milligan G, Tobin AB, Lohse MJ, Hoffmann C: Beta-arrestin biosensors reveal a rapid, receptor-dependent activation/deactivation cycle. Nature (2016) 531(7596):661-664.
5. Butcher AJ, Bradley SJ, Prihandoko R, Brooke SM, Mogg A, Bourgognon JM, Macedo-Hatch T, Edwards JM, Bottrill AR, Challiss RA, Broad LM, Felder, CC, Tobin AB: An antibody biosensor establishes the activation of the m1 muscarinic acetylcholine receptor during learning and memory. J Biol Chem (2016) 291(17):8862-75
6. Bradley SJ, Wiegman CH, Iglesias MM, Kong KC, Butcher AJ, Plouffe B, Goupil E, Bourgognon JM, Macedo-Hatch T, LeGouill C, Russell K Laport, SA, Konig, GM, Kostenis, E, Bouvier M, Chung KF, Amrani Y, and Tobin AB: Mapping physiological g protein-coupled receptor signaling pathways reveals a role for receptor phosphorylation in airway contraction. PNAS (2016) 113(16):4524-4529.
7. Bradley SJ, Tobin AB: Design of next-generation G protein-coupled receptor drugs: Linking novel pharmacology and in vivo animal models. Annual review of pharmacology and toxicology (2016) 56(535-559.
8. Bouzo-Lorenzo M, Santo-Zas I, Lodeiro M, Nogueiras R, Casanueva FF, Castro M, Pazos Y, Tobin AB, Butcher AJ, Camina JP: Distinct phosphorylation sites on the ghrelin receptor, ghsr1a, establish a code that determines the functions of ss-arrestins. Scientific reports (2016) 6(22495.
9. Bolognini D, Tobin AB, Milligan G, Moss CE: The pharmacology and function of receptors for short-chain fatty acids. Mol Pharmacol (2016) 89(3):388-398.
10. Zindel D, Butcher AJ, Al-Sabah S, Lanzerstorfer P, Weghuber J, Tobin AB, Bunemann M, Krasel C: Engineered hyperphosphorylation of the beta2-adrenoceptor prolongs arrestin-3 binding and induces arrestin internalization. Mol Pharmacol (2015) 87(2):349-362
11. Bolognini D, Moss CE, Nilsson K, Petersson AU, Donnelly I, Sergeev E, König GM, Kostenis E, Kurowska-Stolarska M, Miller A, Dekker N, Tobin AB, Milligan G. A Novel Allosteric Activator of Free Fatty Acid 2 Receptor Displays Unique Gi-functional Bias. (2016) J. Biol. Chem. 291(36):18915-18931
12. Rossi M, Ruiz de Azua I, Barella LF, Sakamoto W, Zhu L, Cui Y, Lu H, Rebholz H, Matschinsky FM, Doliba NM, Butcher AJ, Tobin AB, Wess J: CK2 acts as a potent negative regulator of receptor-mediated insulin release in vitro and in vivo. PNAS (2015) 112(49):E6818-6824.
13. Doerig C, Rayner JC, Scherf A, Tobin AB: Post-translational protein modifications in malaria parasites. Nature reviews Microbiology (2015) 13(3):160-172.
14. Alam MM, Solyakov L, Bottrill AR, Flueck C, Siddiqui FA, Singh S, Mistry S, Viskaduraki M, Lee K, Hopp CS, Chitnis CE, Doerig C, Moon RW, Green JL, Holder AA, Baker DA and Tobin AB: Phosphoproteomics reveals malaria parasite protein kinase g as a signalling hub regulating egress and invasion. Nature communications (2015) 6 p7285.
15. Solyakov L, Halbert J, Alam MM, Semblat JP, Dorin-Semblat D, Reininger L, Bottrill AR, Mistry S, Abdi A, Fennell C, Holland Z, Demarta C, Bouza Y, Sicard A, Nivez MP, Eschenlauer S, Lama T, Thomas DC, Sharma P, Agarwal S, Kern S, Pradel G, Graciotti M, Tobin AB, Doerig C: Global kinomic and phospho-proteomic analyses of the human malaria parasite plasmodium falciparum. Nature communications (2011) 2(565.
16. Poulin B, Butcher A, McWilliams P, Bourgognon JM, Pawlak R, Kong KC, Bottrill A, Mistry S, Wess J, Rosethorne EM, Charlton SJ, Tobin AB: The m3-muscarinic receptor regulates learning and memory in a receptor phosphorylation/arrestin-dependent manner. PNAS (2010) 107(20):9440-9445.
17. Kong KC, Butcher AJ, McWilliams P, Jones D, Wess J, Hamdan FF, Werry T, Rosethorne EM, Charlton SJ, Munson SE, Cragg HA, Smart AD, Tobin AB: M3-muscarinic receptor promotes insulin release via receptor phosphorylation/arrestin-dependent activation of protein kinase D1. PNAS (2010) 107(49):21181-21186.
18. Tobin AB, Butcher AJ, Kong KC: Location, location, location...Site-specific gpcr phosphorylation offers a mechanism for cell-type-specific signalling. Trends in pharmacological sciences (2008) 29(8):413-420.
19. Torrecilla I, Spragg EJ, Poulin B, McWilliams PJ, Mistry SC, Blaukat A, Tobin AB: Phosphorylation and regulation of a g protein-coupled receptor by protein kinase CK2. The Journal of cell biology (2007) 177(1):127-137.
20. Tobin AB, Nahorski SR: Rapid agonist-mediated phosphorylation of m3-muscarinic receptors revealed by immunoprecipitation. The Journal of biological chemistry (1993) 268(13):9817-9823.

