Dr Carmen Huesa
- Senior Lecturer (Immunology & Infection)
Biography
I received my BSc hons (1997-2002) with industrial placement (Novartis, Basel, Switzerland) from the University of Aberdeen. After two rewarding years working on antibody engineering at a small spin-out company, Haptogen Ltd., I completed my Ph.D on the mechanotransduction of cells of the osteoblast lineage (2009), also at the University of Aberdeen, under the supervision of Professor Miep Helfrich and Professor Richard Aspden.
My interest in all things bone has allowed me to apply the knowledge gained during my formative years to other aspects of bone. At the Roslin Institute with Professor Colin Farquharson, we investigated the role of Phospho1 in bone mineralisation and discovered the role of Phospho1 in energy metabolism. This interest in energy metabolism led me to a project focusing on the role of eNPP1 with Dr Vicky McRae.
Wanting to return to my interest in the mechanical stimulation of bone and how this may influence disease I joined a multidisciplinary group at the University of Glasgow, led by Professor Bill Ferrell, Professor John Lockhart and Professor Carl Goodyear to investigate the role of Protease Activated Receptor 2 in Osteoarthritis.
I moved to the University of Edinburgh to investigate osteoblast interaction with prostate cancer cells in bone metastasis with Dr Bin Zhi Qian. I then obtained an Early Career Fellowship from Versus Arthritis to investigate pain in osteoarthritis and returned to the University of Glasgow. This research led me to my current research interest on the role of bone in osteoarthritis, which is currently funded by a Career Development Fellowship from Versus Arthritis.
![]() Follow me on Twitter: @CHuesa
Follow me on Twitter: @CHuesa
Research interests
Osteoarthritis is a disease of the degenerating joint and as such it does not develop overnight. For example, osteoarthritis can develop after a severe joint injury, but the lack of mobility and painful stages of the disease only appear years or even decades after the initial injury. Despite the link between joint injury and subsequent development of disease there are no available treatments to prevent or delay severe disease. Our data indicates that bone has a fundamental impact on the later stages of disease. However, it remains to be determined how bone exerts this effect. Understanding how bone interacts with the cartilaginous cushion that ensures joint mobility at the molecular level will offer insight into osteoarthritis progression in the crucial late stages of the disease. This greater knowledge of the disease will likely lead to the identification of new targets to use for future treatments that will help prevent, or at least delay, late-stage osteoarthritis.
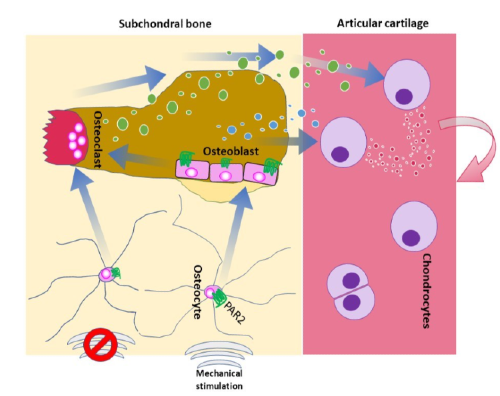
Although the idea that bone regulates osteoarthritis development is not new, it has not yet been determined how exactly this is happening. The novelty of this approach lies in how we are looking at bone, because we are not studying the bone itself, but more how it communicates with its surroundings. Thus, we aim to examine the signals emanating from bone rather than the characteristics of the bone.
Another research interest focuses around the development of peripheral pain in late stage osteoarthritis and the role that biological sex may have.
Over the age of 60, women are twice as likely to develop OA, highlighting that biological sex is one of the most common risk factors. The mechanisms underlying the differences between men and women with OA are unknown; however, hormonal differences are obvious candidates. Before the age of 50, the prevalence of OA in men is greater than in women, conversely, there is an increased risk of OA development amongst post-menopausal women, which has been linked with a reduction is oestrogen, one of the female sex hormones. The decrease in oestrogen in post-menopausal women has been shown to affect the tissues that make the joint. In addition to this, menopause and consequent decrease in oestrogen has been linked to pain. In the clinic it has become difficult to untangle the relationship between sex hormones and symptomatic OA due to the large variability of influencing factors such as years from menopause, type of menopause and type of hormone replacement therapy.
Funding
Grants
Grants and Awards listed are those received whilst working with the University of Glasgow.
- Understanding the communication between the osteoblast lineage and articular cartilage chondrocytes.
Versus Arthritis
2022 - 2027
- Osteoarthritic pain in a 'postmenopausal' murine model
Tenovus Scotland
2021 - 2021
Supervision
Content to follow ...
Public Engagement
We take part in public engagement events to share our enthusiasm for science with a set of entertaining experiments that show how amazing bone is and how important it is for our health. Last year, we took part in the ARCadia festival here at the University of Glasgow and loved the interaction with attendees.
The series of videos embeded below allow us to show our public engagement activities to a broader audience, so feel free to view and share.
If you are keen to have one of our group perform these activities in your school, ask your science teacher to get in touch with me (Carmen.Huesa@glasgow.ac.uk) and we will happily come around with our cool experiments to show how amazing bone is.
THE AMAZING PROPERTIES OF BONE (THE BIG WEEKEND)
Dr Carmen Huesa, Maria Laura Vieri, and Patricia Riedlova prepared activities to show what an amazing tissue bone is.
Part 1 | Bone Density three-point bending
Part 2 | Tower Drop Bone Density
Part 3 | Trabecular Structure Bone Density





