Reducing neurotoxicity in childhood ALL
Most children with acute lymphoblastic leukaemia (ALL) are cured, but treatment is arduous, intensive and causes significant acute and long-term side-effects.
Neurological toxicity is common: about 5-10% of patients suffer an acute neurological serious adverse event and about one-third of survivors have neurocognitive impairment. Despite this, risk-factors for developing neurotoxicity are poorly understood.
To improve neurological outcomes, it is important to routinely evaluate the relative neurotoxicity rates of different treatment approaches, identify individuals at highest risk, and devise evidence-based interventions to reduce or eliminate this disabling complication.
We are currently trying to find out why some children get neurotoxicity and others don’t, as well as whether we can intervene to reduce the occurrence or severity of neurotoxicity. Projects running in the lab include:
 BioCAN
BioCAN
The Biomarkers and the discovery of new therapeutic targets for Chemotherapy Associated Neurotoxicity study asks whether simple computer-based programmes, genetic or cerebrospinal fluid (CSF) tests can identify children at risk of brain complications.
In addition, it is investigating whether neurotoxicity is related to a build-up of certain chemicals in the brain that could be blocked by new treatments.
If we can predict patients at risk, we might be able to design interventions (such as brain training programmes or new drugs) to reduce or prevent the damaging effects of chemotherapy on the brain.
 BRAIN
BRAIN
The BRAIN (Biomarkers to Reform Approaches to Therapy-Induced Neurotoxicity) study will identify patient and treatment risk-factors for chemotherapy-associated neurotoxicity, and prospectively identify children requiring neuropsychological intervention. It uses state-of-the-art neurocognitive assessment tools, novel toxicity reporting systems and harnesses the unique statistical power of one of the world’s largest childhood leukaemia trials – ALLTogether1. BRAIN is an approved ALLTogether1 sub-study, developed by a UK team of experts in CNS leukaemia (Halsey), Neuropsychology (Thomas), ALL trials (Moppett, Clifton-Hadley) and Biostatistics (Kirkwood).
Aims:
- To assess the cumulative neurotoxicity of each randomized treatment arm,
- To identify children with neurocognitive impairment for neuropsychology referral
- To develop accurate risk prediction models for adverse neurocognitive outcomes, enabling future targeted interventions.
All children in the ALLTogether trial will be eligible to participate. Acute neurotoxic events will be electronically captured during treatment, chronic neurocognitive impairment will be measured using patient friendly CogState software at the end of treatment. Results will be correlated with treatment and demographic variables to identify risk-factors for adverse outcomes. Results will also be fed into related ALLTogether1 studies using genome-wide association studies for key treatment related toxicities and linked to patient reported outcome measures.
The results of BRAIN will transform our understanding of this serious treatment complication and enable future trials of interventions targeted to at-risk individuals.
The Ponte di Legno Deep Phenotyping-Genotyping Neurotoxicity study
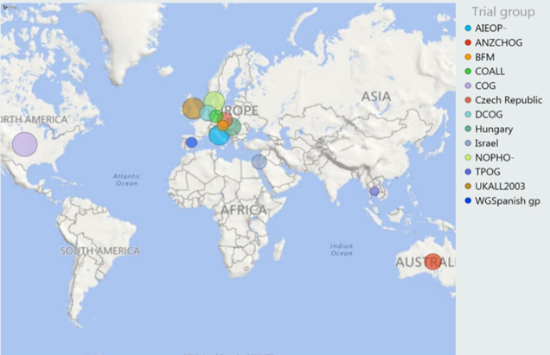
Acute neurotoxicity events, such as stroke-like syndrome/posterior reversible encephalopathy syndrome, remain devastating but rare complications of childhood acute lymphoblastic leukaemia (ALL) treatment. The risk factors and predictors of outcome for these conditions are largely unknown because each individual national trial group sees too few cases to enable in-depth analysis. To tackle this, an international consortium has been formed comprising fourteen national trial groups representing over 20 countries enabling information to be gathered on >1800 cases worldwide.
This project aims to:
- Carry out deep phenotyping of these cases to identify clinical features, risk factors and predictors of poor outcome.
- Genotype a discovery cohort of severe cases to identify risk alleles for development of neurotoxicity
- Create and analyse a validation cohort to test whether identified risk alleles can predict neurotoxicity in independent cohorts of children with ALL.
This information is predicted to improve outcome for these patients by:
- identifying modifiable acquired risk factors (such as drug interactions),
- improved prediction of outcomes enabling improved counselling of affected families and provision of appropriate supportive care and
- identifying genetic risk alleles that may enable precision medicine by tailoring therapies according to an individual’s risk of toxicity.
Raising Clinical Awareness
In addition, to these ongoing projects we have published a number of papers to raise awareness of drug interactions and appropriate drug dosing to maximise effectiveness and minimise toxicity of our current CNS-directed therapy.
Publications:
1. Forster VJ, Bell G, Halsey C. Should nitrous oxide ever be used in oncology patients receiving methotrexate therapy? Paediatr Anaesth. 2020;30(1):9-16.
2. Forster VJ, van Delft FW, Baird SF, Mair S, Skinner R, Halsey C. Drug interactions may be important risk factors for methotrexate neurotoxicity, particularly in pediatric leukemia patients. Cancer chemotherapy and pharmacology. 2016;78(5):1093-1096.
3. Forster VJ, van Delft FW, Baird SF, Mair S, Skinner R, Halsey C. Reply: Methotrexate neurotoxicity due to drug interactions: an inadequate folinic acid effect. Cancer Chemother Pharmacol. 2017.
4. Schmiegelow K, Attarbaschi A, Barzilai S, et al. Consensus definitions of 14 severe acute toxic effects for childhood lymphoblastic leukaemia treatment: a Delphi consensus. The Lancet Oncology. 2016;17(6):e231-e239.
5. Wilson R, Osborne C, Halsey C. The Use of Ommaya Reservoirs to Deliver Central Nervous System-Directed Chemotherapy in Childhood Acute Lymphoblastic Leukaemia. Paediatric drugs. 2018;20(4):293-301.

