
Welcome to the Cagan laboratory
Our goal is to explore the biology of therapeutics.
 Why do lead compounds work well in model systems but most fail in clinical trials? Our lab is devoted to embracing complexity, both in our models and in the therapeutics we are developing. Oh, and we use mouse and human organoids as well as the fruit fly Drosophila as our starting point.
Why do lead compounds work well in model systems but most fail in clinical trials? Our lab is devoted to embracing complexity, both in our models and in the therapeutics we are developing. Oh, and we use mouse and human organoids as well as the fruit fly Drosophila as our starting point.
 Overview
Overview
We explore two categories of disease: cancer [thyroid, colorectal, breast, lung] and Mendelian (inherited) diseases [RASopathies, Tauopathies]. We use flies and mammalian models (organoids, mouse xenografts) to explore therapeutic mechanisms, to develop new lead compounds, and to execute a fly-to-bedside clinical trial where we treat cancer patients. Incidentally, the art on the left is from Jazz Cagan. Yea, we think so too.
Cancer
 The primary focus of our laboratory is cancer. We contribute to the cancer field in two broad ways. First, we build highly complex Drosophila models designed to capture the genomic complexity of individual human colorectal, thyroid, lung, and breast cancer patients. These ‘fly avatars’ allow us to explore how altering 5-15 genes in a single transgenic animal alters tumor progression and drug response. Our organoid work then builds on our fly findings, validating and extending to explore mammalian-specific aspects.
The primary focus of our laboratory is cancer. We contribute to the cancer field in two broad ways. First, we build highly complex Drosophila models designed to capture the genomic complexity of individual human colorectal, thyroid, lung, and breast cancer patients. These ‘fly avatars’ allow us to explore how altering 5-15 genes in a single transgenic animal alters tumor progression and drug response. Our organoid work then builds on our fly findings, validating and extending to explore mammalian-specific aspects.
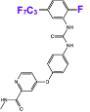 Our second approach is drug development. Working with our close collaborators Arvin Dar, Avner Schlessinger, and their teams, we have developed a novel platform—combining fly genetics with medicinal and computational chemistry—to develop lead compounds that attack the cancer network in the context of the whole animal. These ‘Tumor Calibrated Inhibitors’ represent a new way towards addressing drug liabilities including resistance and toxicity.
Our second approach is drug development. Working with our close collaborators Arvin Dar, Avner Schlessinger, and their teams, we have developed a novel platform—combining fly genetics with medicinal and computational chemistry—to develop lead compounds that attack the cancer network in the context of the whole animal. These ‘Tumor Calibrated Inhibitors’ represent a new way towards addressing drug liabilities including resistance and toxicity.
Fly-To-Bedside Clinical Trial
Based on our work exploring the relationship between genomic complexity and drug resistance, we opened a fly-to-bedside clinical trial as part of the Center for Personalized Cancer Therapeutics, and publishing two fly-to-patient reports.
Mendelian Diseases
RASopathies
 The RASopathies are a family of inherited disorders characterized by mutations that activate the RAS/ERK signaling network. Patients present with a broad palette of developmental and post-natal defects. Currently, only symptomatic care is available for affected individuals. We are working with Bruce Gelb and colleagues to develop 30 new RASopathy models, exploring the biology, and (with Bruce, Jian Jin and colleagues) developing new lead therapeutics using chemical evolution.
The RASopathies are a family of inherited disorders characterized by mutations that activate the RAS/ERK signaling network. Patients present with a broad palette of developmental and post-natal defects. Currently, only symptomatic care is available for affected individuals. We are working with Bruce Gelb and colleagues to develop 30 new RASopathy models, exploring the biology, and (with Bruce, Jian Jin and colleagues) developing new lead therapeutics using chemical evolution.

