Protein antibiotics as novel therapeutic agents for the treatment of chronic biofilm-mediated infection.
Researchers:
Laura Martin l.martin.2@research.gla.ac.uk
Carla Brown c.brown.4@research.gla.ac.uk
The emergence of pan-resistant strains of Gram-negative pathogens and the ability of many bacteria to form multidrug-resistant biofilms during chronic infection poses the grave threat of bacterial infections that are truly untreatable with our current armoury of antibiotics. Despite obvious clinical need, few new antibiotics have entered clinical practice in recent years. For 'difficult to treat' Gram-negative bacteria such as Pseudomonas aeruginosa and Escherichia coli, where the presence of outer membrane and multidrug-efflux pumps severely limit the effectiveness of whole classes of antibiotics, the need is particularly pressing. An alternative approach to antimicrobial treatment is to use the well-characterized species-specific colicin-like bacteriocins which are produced by a wide range of Gram-negative bacteria, including Pseudomonas aeruginosa and Escherichia coli. Our current work on colicin-like bacteriocins aims to determine whether these potent antimicrobial agents are effective at killing bacteria growing in the biofilm state and during infection.
Pseudomonas aeruginosa is an important human pathogen, one of the commonest causes of nosocomial pneumonia and the main respiratory pathogen producing progressive respiratory failure in patients with cystic fibrosis (CF). The ability of P. aeruginosa to form multi-drug resistant biofilms during chronic infection can render this pathogen essentially untreatable with existing antibiotics. This is exemplified in cystic fibrosis patients where, despite aggressive antibiotic therapy, chronic lung infection with P. aeruginosa and the concomitant intense inflammatory response leads to a progressive loss of lung function and is the major proven cause of mortality among this group of patients. Thus, there is a critical need to develop new antibiotics to treat P. aeruginosa infections.
S-type pyocins are known to bind to outer membrane receptors involved in nutrient uptake, such as iron-siderophore receptors, effectively parasitizing host nutrient uptake systems. Our data shows that pyocins are highly active against P. aeruginosa clinical isolates growing in the biofilm state and in a model of infection. These data suggest pyocins may make useful therapeutics against chronic P. aeruginosa infection for which currently available antibiotics are inadequate. 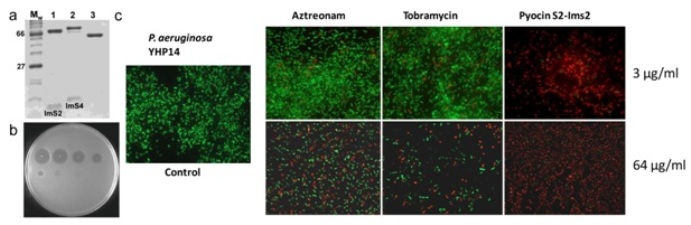
Purification of S-type pyocins and killing activity of the pyocin S2-Im2 complex against P. aeruginosa. (a) SDS-PAGE of purified pyocin S2-ImS2 complex (lane 1). (b) Activity of purified pyocin S2-ImS2 against P. aeruginosa. A 5-fold serial dilution (starting concentration, 2 mg/ml [top left]) of pyocin S2-ImS2 was spotted onto a lawn of growing P. aeruginosa strain YHP17 which was grown overnight at 37°C. Clear zones indicate cell death. (c) Activity of pyocin S2-ImS2 against P. aeruginosa biofilms. Biofilms (24 h) of P. aeruginosa YHP14 were grown on poly-l-lysine glass cover slides, treated with a pyocin S2 at 3 μg/ml and 64 μg/ml for 1 h, and visualized by fluorescence microscopy using LIVE/DEAD cell viability staining. Red, dead cells; green, live cells.
References
Smith K, Martin L, Rinaldi A, Rajendran R, Ramage G, Walker D. (2012) Activity of pyocin S2 against Pseudomonas aeruginosa biofilms. Antimicrob. Agents. Chemother. 56: 1599-601.
Brown CL, Smith K, McCaughey L, Walker D. (2012) Colicin-like bacteriocins as novel therapeutic agents for the treatment of chronic biofilm-mediated infection. Biochem. Soc. Trans. 40: 1549-52.

