Isotope Biochemistry Laboratory
Activities
Carbohydrate metabolism
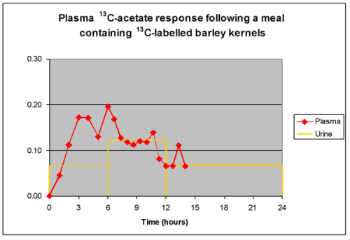 Within our research theme of chronic disease, we have a developing interest in factors controlling the glycaemic response of a meal. Insulin resistance (IR) and a complex array of symptoms that are together called the metabolic syndrome, are associated with lack of control of postprandial glycemia. IR is associated with many chronic inflammatory diseases, including type II diabetes, cardiovascular disease and certain cancers. As partners within the EU Eurostarch project, SIBL contributed to the finding that the short chain fatty acid (SCFA) fermentation products of non-digestible carbohydrates (NDC), produced by the colonic microflora, can moderate the glycaemic response of subsequent meals. Mechanisms by which this can occur and the nutritional management practices and advice that may result are the subject of ongoing research. Our unique ability in this area brings together novel non-invasive methodology to measure SCFA production in vivo with our expertise in compound-specific isotope analysis (Morrison et al., 2004).
Within our research theme of chronic disease, we have a developing interest in factors controlling the glycaemic response of a meal. Insulin resistance (IR) and a complex array of symptoms that are together called the metabolic syndrome, are associated with lack of control of postprandial glycemia. IR is associated with many chronic inflammatory diseases, including type II diabetes, cardiovascular disease and certain cancers. As partners within the EU Eurostarch project, SIBL contributed to the finding that the short chain fatty acid (SCFA) fermentation products of non-digestible carbohydrates (NDC), produced by the colonic microflora, can moderate the glycaemic response of subsequent meals. Mechanisms by which this can occur and the nutritional management practices and advice that may result are the subject of ongoing research. Our unique ability in this area brings together novel non-invasive methodology to measure SCFA production in vivo with our expertise in compound-specific isotope analysis (Morrison et al., 2004).
Lipid metabolism
With colleagues from the Department of Developmental Medicine, University of Glasgow (Professor LT Weaver), we have been developing 13C breath tests and other non-invasive tests for lipid digestion and absorption measurements. The sensitivity and specificity of the 13C-mixed triglyceride breath test for lipase sufficiency in children with cystic fibrosis, has been significantly improved for clinical use (Slater C, Preston T, & Weaver LT, 2006).
Our earlier work on the elevated fatty acid oxidation in cancer patients is also relevant (Selberg et al., 1991). This condition is not strictly characterised by abnormal metabolism, rather fat oxidation is elevated due to reduced intake and a catabolic hormonal profile in the chronic malnutrition of the cancer patient. Our current interests centre on the interaction of fat metabolism with carbohydrate metabolism. The so-called ‘late second-meal effect’ is one aspect, where the properties of one meal can moderate the glycaemic response of subsequent meals. This thought to result from the action of SCFA product of NDC fermentation. To understand this mechanism, we need to study how the body handles SCFA.
Protein metabolism
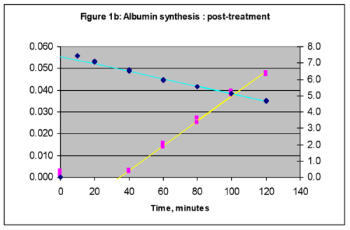 Our early work concerned some of the largest studies of whole body protein turnover in weight-losing cancer patients (Fearon et al., 1988, 1991). We were one of the first to link elevated whole body protein turnover with ongoing inflammatory disease. The simple methodology applied an oral bolus of 15N-glycine with a measure of its dilution in a urinary endproduct, integrated over 24 hours. Although the model has some questionable assumptions, namely concerning the use of a single amino acid to measure protein turnover, experience of its use informed our recent development of the protocol to measure acetate and propionate production. In an attempt to discover the basis of the elevated protein synthesis, we moved on to develop methodologies to measure tissue protein synthesis and finally, the synthetic rate of individual proteins. The former method is limited to the availability of tissue biopsies. The latter method is ideally suited to measure individual plasma proteins. It has benefited from recent improvements in measurement sensitivity.
Our early work concerned some of the largest studies of whole body protein turnover in weight-losing cancer patients (Fearon et al., 1988, 1991). We were one of the first to link elevated whole body protein turnover with ongoing inflammatory disease. The simple methodology applied an oral bolus of 15N-glycine with a measure of its dilution in a urinary endproduct, integrated over 24 hours. Although the model has some questionable assumptions, namely concerning the use of a single amino acid to measure protein turnover, experience of its use informed our recent development of the protocol to measure acetate and propionate production. In an attempt to discover the basis of the elevated protein synthesis, we moved on to develop methodologies to measure tissue protein synthesis and finally, the synthetic rate of individual proteins. The former method is limited to the availability of tissue biopsies. The latter method is ideally suited to measure individual plasma proteins. It has benefited from recent improvements in measurement sensitivity.
Nutritional assessment
Body composition analysis is the most common form of nutritional assessment. The underlying two-compartment model is of body mass comprising body fat and lean body mass or fat free mass:
Body Weight = Body Fat + Fat Free Mass
There are numerous rather crude and inexact methods of body composition analysis that require validation using a primary method. In contrast, there are only three methods that can be termed primary methods: elemental analysis; densitometry and total body water analysis (TBW). Glasgow University has a long tradition of nutritional assessment. Professor Keith Boddy, formerly of the Health Physics and Nuclear Medicine unit at SURRC, pioneered the analysis of Total Body Potassium and multi-elemental analysis using in vivo neutron activation analysis; Professor John Durnin, of the Institute of Physiology, developed apparatus for densitometric analysis and pioneered the secondary method of body fat analysis, skinfold thickness. TBW can be measured with great accuracy using stable isotope dilution. SIBL has developed protocols and maintains instrumentation for TBW analysis. TBW is the only primary body composition method that can be conducted remotely from a laboratory facility (on a hospital ward; in the field).
 Functional measurements in human metabolism are predicted to be a major growth area in the future. Isotope dilution analysis of TBW is directly related to functional studies. In every TBW measurement using hydrogen isotopes, account has to be made of a small proportion of tracer that is incorporated into organic molecules both through exchange and biosynthesis. When using deuterated water as tracer, some 4% is incorporated in proteins and lipids. This is the basis of methods that have been developed to measure protein synthesis, lipogensesis and cholesterogenesis. Further, total energy expenditure and breast milk intake can be quantified by following the elimination of body water labelled with stable isotopes.
Functional measurements in human metabolism are predicted to be a major growth area in the future. Isotope dilution analysis of TBW is directly related to functional studies. In every TBW measurement using hydrogen isotopes, account has to be made of a small proportion of tracer that is incorporated into organic molecules both through exchange and biosynthesis. When using deuterated water as tracer, some 4% is incorporated in proteins and lipids. This is the basis of methods that have been developed to measure protein synthesis, lipogensesis and cholesterogenesis. Further, total energy expenditure and breast milk intake can be quantified by following the elimination of body water labelled with stable isotopes.
Energy expenditure
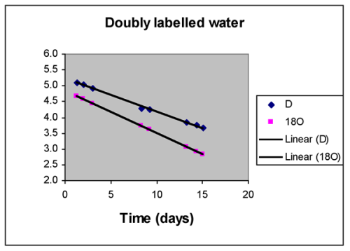 Nutrition and lifestyle factors underlie most chronic diseases. An individual’s energy balance is fundamental. Typically, we take in 50% more calories than required for basal metabolism as physical activity level burns the additional fuel. That is, Physical Activity Level is 1.5 (PAL, defined as Total Energy Expenditure/Resting Energy Expenditure). An endurance athlete will need many more calories from the diet (over 2-fold) whereas a sedentary elderly subject will need considerably less. Should dietary intake exceed TEE, body fat will be laid down. Should diet not meet requirements, body fat stores will be oxidised. We are using PAL as an objective means of assessing lifestyle in cachectic cancer patients, providing a much clearer picture than lifestyle questionnaires. Such patients show very low PAL (~1.3), appearing to adapt to the twin problems of reduced dietary intake and increased REE, by reducing physical activity. We use longitudinal measurements of PAL as a means of assessing the efficacy of nutritional and anti-inflammatory intervention (Moses et al., 2004). We use the doubly labelled water technique to measure TEE. Here, both 2H- and 18O-lablled water are used together. As 18O in water also equilibrates with CO2, it is eliminated from the body as water and carbon dioxide; whereas 2H is only eliminated as water. The difference in elimination rate of the two tracers provides an accurate estimate of total CO2 output and hence, TEE. This method can be applied in free living subjects, typically over two week periods. It is non-hazardous and non-invasive, so it can be applied in children. REE is measured using gas exchange (ventilated hood indirect calorimetry). SIBL has developed protocols and has excellent facilities for measuring TEE funded with MRC JREI support. The same grant funded an indirect calorimeter, which is currently installed within the Wellcome Trust Clinical Research Facility at the Western General Infirmary, Edinburgh. Recent work has applied miniature activity meters (developed by a Glasgow company) to examine the pattern of energy expenditure in patients on a minute-by-minute basis over a one week period. It seeks to validate their use against doubly labelled water.
Nutrition and lifestyle factors underlie most chronic diseases. An individual’s energy balance is fundamental. Typically, we take in 50% more calories than required for basal metabolism as physical activity level burns the additional fuel. That is, Physical Activity Level is 1.5 (PAL, defined as Total Energy Expenditure/Resting Energy Expenditure). An endurance athlete will need many more calories from the diet (over 2-fold) whereas a sedentary elderly subject will need considerably less. Should dietary intake exceed TEE, body fat will be laid down. Should diet not meet requirements, body fat stores will be oxidised. We are using PAL as an objective means of assessing lifestyle in cachectic cancer patients, providing a much clearer picture than lifestyle questionnaires. Such patients show very low PAL (~1.3), appearing to adapt to the twin problems of reduced dietary intake and increased REE, by reducing physical activity. We use longitudinal measurements of PAL as a means of assessing the efficacy of nutritional and anti-inflammatory intervention (Moses et al., 2004). We use the doubly labelled water technique to measure TEE. Here, both 2H- and 18O-lablled water are used together. As 18O in water also equilibrates with CO2, it is eliminated from the body as water and carbon dioxide; whereas 2H is only eliminated as water. The difference in elimination rate of the two tracers provides an accurate estimate of total CO2 output and hence, TEE. This method can be applied in free living subjects, typically over two week periods. It is non-hazardous and non-invasive, so it can be applied in children. REE is measured using gas exchange (ventilated hood indirect calorimetry). SIBL has developed protocols and has excellent facilities for measuring TEE funded with MRC JREI support. The same grant funded an indirect calorimeter, which is currently installed within the Wellcome Trust Clinical Research Facility at the Western General Infirmary, Edinburgh. Recent work has applied miniature activity meters (developed by a Glasgow company) to examine the pattern of energy expenditure in patients on a minute-by-minute basis over a one week period. It seeks to validate their use against doubly labelled water.
Enhanced Recovery After Surgery (ERAS) is a recent initiative that has been given a high priority by health services. Pioneered in Europe, and co-ordinated from Edinburgh, a series of nutritional, surgical and clinical management procedures have been introduced to reduce the effects of surgical trauma. Overall this reduces hospital stay significantly, which is of great benefit to health budgets. With colleagues in Edinburgh, we are applying activity meters to assess physical recovery after surgery. We are also applying metabolic and functional measurements, including measuring liquid phase gastric emptying to assess the assimilation of sip feeds.
Biological synthesis of novel tracers
 Principally through EU funding (www.eurostarch.org), we have built up the capability to produce significant quantities of 13C-labelled grain for nutrition studies. Intrinsic labelling allows us to produce complex metaprobes that can be processed and incorporated into meals, behaving exactly as normal foodstuffs. The alternative, taking glucose with a starch based meal, does not faithfully trace the rate and extent of digestion of that meal. Using greenhouse and growth chamber facilities at the Scottish Agricultural College, Edinburgh, we have produced kg quantities of 13C-labelled barley and Durum wheat grain. These have been processed and incorporated into foodstuffs to investigated carbohydrate digestion and fermentation, in collaboration with our EU partnership. We have also gained experience in producing soft wheat and hay, the latter as a potential cattle feed to label milk products. We also have previous experience of biosynthesis of 15N, 13C and 2H-labelled microbes.
Principally through EU funding (www.eurostarch.org), we have built up the capability to produce significant quantities of 13C-labelled grain for nutrition studies. Intrinsic labelling allows us to produce complex metaprobes that can be processed and incorporated into meals, behaving exactly as normal foodstuffs. The alternative, taking glucose with a starch based meal, does not faithfully trace the rate and extent of digestion of that meal. Using greenhouse and growth chamber facilities at the Scottish Agricultural College, Edinburgh, we have produced kg quantities of 13C-labelled barley and Durum wheat grain. These have been processed and incorporated into foodstuffs to investigated carbohydrate digestion and fermentation, in collaboration with our EU partnership. We have also gained experience in producing soft wheat and hay, the latter as a potential cattle feed to label milk products. We also have previous experience of biosynthesis of 15N, 13C and 2H-labelled microbes.
Chemical synthesis of novel tracers
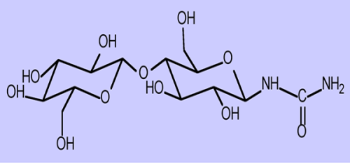 A number of simple metaprobes are not yet available from commercial sources. We have worked closely with commercial suppliers of such tracers to address this, and have also undertaken synthetic work to address this deficiency. We have an interest in controlled tracer delivery to the bowel. Existing technologies such as coated capsules, are inaccurate and not easily applied to tracer studies. We have experience of chemical synthesis of two classes of compounds that can be used to deliver metaprobes to the colon: glycosyl-ureides and carbohydrate esters. The former class is not metabolised to any degree by human digestive enzymes but when taken orally, they are hydrolysed by colonic bacteria. They can be used to measure oro-caecal transit (13C labelled urea is released and hydrolysed to CO2 in the caecum; which appears in breath) or to deliver 15N-ammonium, 13C-CO2 or 13C-glucose to the colon. The second class of compounds can be applied to deliver known quantities of 13C-labelled SCFA to the colon, which can serve as tracers for SCFA production studies.
A number of simple metaprobes are not yet available from commercial sources. We have worked closely with commercial suppliers of such tracers to address this, and have also undertaken synthetic work to address this deficiency. We have an interest in controlled tracer delivery to the bowel. Existing technologies such as coated capsules, are inaccurate and not easily applied to tracer studies. We have experience of chemical synthesis of two classes of compounds that can be used to deliver metaprobes to the colon: glycosyl-ureides and carbohydrate esters. The former class is not metabolised to any degree by human digestive enzymes but when taken orally, they are hydrolysed by colonic bacteria. They can be used to measure oro-caecal transit (13C labelled urea is released and hydrolysed to CO2 in the caecum; which appears in breath) or to deliver 15N-ammonium, 13C-CO2 or 13C-glucose to the colon. The second class of compounds can be applied to deliver known quantities of 13C-labelled SCFA to the colon, which can serve as tracers for SCFA production studies.
Non-invasive 13C breath tests
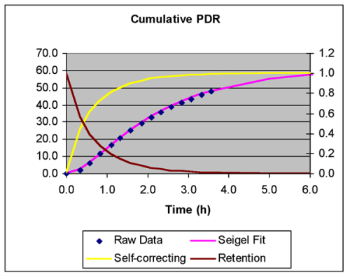 The mass spectrometric instrumentation used for non-invasive breath tests was first developed within SIBL and we are equipped with the latest equipment. We continue to develop non-invasive breath tests when they can be of use for our collaborative medical and veterinary research. The 13C urea breath test for diagnosing H. pylori infection is most widely applied. We no longer apply this test routinely, having commissioned the facility at the Royal Hospital for Sick Children, Glasgow. We serve to back up and calibrate this facility and that within Glasgow Royal Infirmary and to train visiting scientist in this procedure. Our main area of interest is to develop and apply functional tests of gastrointestinal motility, macronutrient digestion and organ function. We have developed protocols to measure liquid or solid phase gastric emptying, oro-caecal transit time, fat digestion and liver function. We are currently applying the 1-13C-acetate breath test to measure liquid phase gastric emptying as a means to study the assimilation of sip feed supplements.
The mass spectrometric instrumentation used for non-invasive breath tests was first developed within SIBL and we are equipped with the latest equipment. We continue to develop non-invasive breath tests when they can be of use for our collaborative medical and veterinary research. The 13C urea breath test for diagnosing H. pylori infection is most widely applied. We no longer apply this test routinely, having commissioned the facility at the Royal Hospital for Sick Children, Glasgow. We serve to back up and calibrate this facility and that within Glasgow Royal Infirmary and to train visiting scientist in this procedure. Our main area of interest is to develop and apply functional tests of gastrointestinal motility, macronutrient digestion and organ function. We have developed protocols to measure liquid or solid phase gastric emptying, oro-caecal transit time, fat digestion and liver function. We are currently applying the 1-13C-acetate breath test to measure liquid phase gastric emptying as a means to study the assimilation of sip feed supplements.
We also have experience of 13C breath tests of liver function. We have applied caffeine and erythromycin, both substrates being demethylated through different enzymes within the microsomal Cytochrome P450 complex. We are currently applying the 13C-galactose breath test in dogs with suspect liver function, having first applied it in human subjects undergoing liver resection. We also have experience of simple gas analysis in breath for functional studies (hydrogen; hydrocarbons; CO; NO) and in carbohydrate analysis in body fluids for non-invasive measurement of gut permeability.
 In undertaking our metabolic studies using 13C, whether for macronutrient digestion or SCFA production, we frequently measure the rate and extent of substrate oxidation. In addition to accurate measurements of 13C enrichment in breath CO2, we frequently need to estimate CO2 output (VCO2). Here, our experience of measuring energy expenditure and applying individual heart rate monitors, has allowed us to control VCO2 during breath tests. We have validated this approach using 13C and 2H tracers simultaneously (Slater et al., 2006). In collaboration with the MRC HNR laboratories in Cambridge, we have also validated the use of a new mathematical model to derive accurate kinetic outcomes by correcting breath tests for the delay in appearance of 13CO2 due to the slow turnover of body’s large CO2 pool. Together these advances overcome limitations to the clinical application of 13C breath tests, which can now produce kinetic data that is comparable to that from radioscintigraphy.
In undertaking our metabolic studies using 13C, whether for macronutrient digestion or SCFA production, we frequently measure the rate and extent of substrate oxidation. In addition to accurate measurements of 13C enrichment in breath CO2, we frequently need to estimate CO2 output (VCO2). Here, our experience of measuring energy expenditure and applying individual heart rate monitors, has allowed us to control VCO2 during breath tests. We have validated this approach using 13C and 2H tracers simultaneously (Slater et al., 2006). In collaboration with the MRC HNR laboratories in Cambridge, we have also validated the use of a new mathematical model to derive accurate kinetic outcomes by correcting breath tests for the delay in appearance of 13CO2 due to the slow turnover of body’s large CO2 pool. Together these advances overcome limitations to the clinical application of 13C breath tests, which can now produce kinetic data that is comparable to that from radioscintigraphy.
The gut microbiome
In 1958, Meselson and Stahl published their seminal work describing the semi-conservative replication of DNA (Meselson & Stahl, 1958). They grew yeast cells on highly enriched 15N, using analytical ultracentrifuge to demonstrate the labelling pattern in isolated DNA. To this day, analytical ultracentrifugation can only separate (detect) very high stable isotope enrichments (~20 atom % excess and greater). This same approach was recently re-introduced in microbial ecology by Inerson and coworkers (Radajewski et al., 2000) to demonstrate the most active members of the microbial community when subjected to a highly enriched energy source. These authors went on to use modern microbial genetics techniques (PCR with sequence analysis) to determine the genotype of the most active species. This approach was termed Stable Isotope Probing (SIP). We are introducing SIP to explore the gut microbiome, which is every bit as complex as the soil microbiome. In place of analytical untracentrifugation, we are developing IRMS in combination with nucleic acid probing to greatly increase the sensitivity of this approach (MacGregor et al., 2002) so that they may be applied to real world studies of the human gut microbiome. Studies of the microbial community structure are entirely compatible with studies of the combined products of microbial processes (the gut metabolome).
Hazardous endogenous products
We continue to research the mechanism of endogenous formation on N-nitrosamines. This research on the mechanism of gastric cancer continues with Professor KEL McColl and colleagues of Glasgow Western Infirmary. We are applying GCMSn techniques to monitor the formation of volatile nitrosamines in aqueous and lipid based systems. We exploit 15N tracers in vivo in order to explore the mechanism of N-nitrosoamine formation at the gastro-oesophageal junction (Winter et al., 2007). We also use 2H-labelled internal standards for accurate quantitation. Using an in vitro model stomach, we are investigating the role of antioxidant vitamins and dietary polyphenols in attenuating nitrosation. We are also investigating bile salts as potential nitrosatable compounds.
There may well be linkages between our studies of endogenous nitrosation and gastric cancer and dietary causes of colon cancer. The EPIC study demonstrated the association of red meat intake, assumed to be a rich source of nitrosatable amines, faecal markers of endogenous nitrosation and the prevalence of bowel disease. Dietary and endogenous proteins which escape digestion are fermented in the bowel, producing a number of products. Using GCMS, we are investigating urinary branched chain fatty acids (fermentation products of branched chain amino acids) and phenols (from tyrosine) as indicators of this process. Some amine and amide products of protein fermentation are bioactive and some may be nitrosatable. Such products of protein fermentation may be detrimental to bowel health.
Functional metabolomics
The term metabolomics is used to describe the global analysis of small molecules or metabolites in living systems. Functional metabolomics concerns measurement and interpretation of the flux of small molecules. The study of small molecules encompasses all their sources and sinks, including the degradation and synthesis of macromolecules, such as proteins and nucleic acids. Functional metabolomics can be looked upon as an extension of functional proteomics and other 'omics'. There is a growing consensus that metabolomics is set to become essential to our understanding of body function in health and disease, over and above the insights provided by proteomics and genomics. It should be noted that this definition of functional metabolomics includes applications in human, veterinary, animal, plant and microbial systems. Macronutrient and intermediary metabolism are key, but micronutrient metabolism (vitamins; antioxidants; minerals) is not excluded and can also be studied using stable isotope tracers (metaprobes). Metaprobes allow unique and unambiguous measurements of the concentration and flux of molecules. The science of functional metabolomics is at a very early stage of development. Developments in novel tracers, instrumentation, protocols and bioinformatics are all necessary before functional metabolomics (metabolite flux measurement) can simultaneously encompass the range of small molecules that metabolomics (metabolite concentration) aspires to measure. There is no doubt that mass spectrometry and NMR instrumentation will be used. Within SIBL, we intend to pioneer and establish mass spectrometric instrumentation, working with the SUERC consortium universities to provide a complete solution to facilitate functional metabolomics studies. To this aim, we are currently seeking external support to fund a triple quadruple LC-MSMS instrument as a structural analysis tool to compliment our new LC-IRMS facility.
Instrumental developments
SIBL pioneered CF-IRMS, at first developing this for bulk analysis of 15N and 13C tracers in environmental biology. We then adapted it for medical research, firstly for protein turnover studies. It was the need to measure amino acid oxidation that caused us to develop the technique for analysis of 13C in breath CO2. This approach quickly became the standard method for 13C breath tests, which was set to become the largest single market worldwide for IRMS instruments. We went on to automate and develop the approach for 18O analysis in biological fluids, which has become the commonest analytical technique for doubly labelled water studies. We are also equipped with CF-IRMS instrumentation for 2H tracer studies. We extended gas analysis by CF-IRMS to 15N in N2 for environmental nitrogen fixation and denitrification studies, and to 13C analysis in CH4 for fermentation studies. SIBL maintains a large variety of instruments for bulk and compound-specific isotope analysis.
C and N biogeochemistry
 Over the years SIBL has taken part in UK and international studies, principally related to the carbon and nitrogen biogeochemistry in aquatic environments. We have introduced a number of new stable isotope techniques to quantify the turnover of particulate and dissolved organic matter in marine systems and freshwaters. Indeed, SIBL conceived and developed CF-IRMS with such applications in mind. Through our participation in the NERC-funded BOFS programme, part of the UK contribution to JGOFS, and using nitrogen isotope techniques, we studied the drawdown of atmospheric carbon in the North Atlantic and South Atlantic. In this way, we contributed to improved international estimates of atmospheric carbon dioxide drawdown by the oceans. Applications in ecology are one of the largest growth areas of stable isotopes.
Over the years SIBL has taken part in UK and international studies, principally related to the carbon and nitrogen biogeochemistry in aquatic environments. We have introduced a number of new stable isotope techniques to quantify the turnover of particulate and dissolved organic matter in marine systems and freshwaters. Indeed, SIBL conceived and developed CF-IRMS with such applications in mind. Through our participation in the NERC-funded BOFS programme, part of the UK contribution to JGOFS, and using nitrogen isotope techniques, we studied the drawdown of atmospheric carbon in the North Atlantic and South Atlantic. In this way, we contributed to improved international estimates of atmospheric carbon dioxide drawdown by the oceans. Applications in ecology are one of the largest growth areas of stable isotopes.
Our current interest in the potential of CSIA in amino acids links directly to the many bulk isotope studies from ecology undertaken by the NERC Life Sciences Mass Spectrometry Facility Node at SUERC Firstly, we are progressing with developing LC-IRMS analysis of amino acids, which we wish to see applied to ecological studies. We predict that 13C analysis of the carbon skeleton of key non-essential amino acids in tissue protein will illustrate the switch between dietary and stored energy sources in times of stress. This is especially true of aquatic organisms, where a switch to use of protein as the main energy source would predict starvation (the carbon skeletons of aspartate and glutamate reflect the precursors of their respective TCA cycle keto acids, oxaloacetete and a-keto-glutarate). The 15N signature of amino acids should provide a greater insight into trophic relationships than bulk protein 15N signature as well as a clearer picture of nutritional stress (Preston & Slater, 1994).
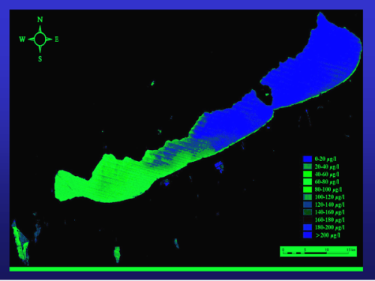 Ongoing collaborative studies continue in one of Europe’s largest freshwater lakes, Lake Balaton in Hungary. Extensive engineering works in the 1980s greatly reduced nitrogen and phosphorous loading into this important lake. In terms of phosphorous loading, its trophic status improved from eutrophic to mesotrophic. We predicted that primary production in the summer would move from being limited by phosphorous supply to being limited by nitrogen. In the summer months water quality can be threatened by cyanobacterial blooms. We helped to establish 15N techniques which have been used to monitor nitrogen cycle processes in Lake Balaton. The summer phytoplankton has indeed become dominated by heterocystous (nitrogen fixing) cyanobacteria. The hydrobiology laboratory at the Balaton Limnological Research Institute, remains one of a very few in the world equipped to study nitrogen cycling processes using 15N methodology, including gaseous nitrogen fixation and denitrification. Current work is aimed at informing water management authorities to continue to improve water quality.
Ongoing collaborative studies continue in one of Europe’s largest freshwater lakes, Lake Balaton in Hungary. Extensive engineering works in the 1980s greatly reduced nitrogen and phosphorous loading into this important lake. In terms of phosphorous loading, its trophic status improved from eutrophic to mesotrophic. We predicted that primary production in the summer would move from being limited by phosphorous supply to being limited by nitrogen. In the summer months water quality can be threatened by cyanobacterial blooms. We helped to establish 15N techniques which have been used to monitor nitrogen cycle processes in Lake Balaton. The summer phytoplankton has indeed become dominated by heterocystous (nitrogen fixing) cyanobacteria. The hydrobiology laboratory at the Balaton Limnological Research Institute, remains one of a very few in the world equipped to study nitrogen cycling processes using 15N methodology, including gaseous nitrogen fixation and denitrification. Current work is aimed at informing water management authorities to continue to improve water quality.
* Cyanobacterial Chlorophyll a in Lake Balaton: satellite image courtesy of Dr AN Tyler, Stirling University.
Provenance studies
Bulk analysis of 13C, 15N, 2H & 18O abundance individually or in combinations, has proven a unique and invaluable tool in provenance studies in areas such as food adulteration, drugs of abuse and forensic science. The common theme in these applications is the use of natural abundance measurements to investigate source terms and to generate high quality data that can be defended in court, if necessary. The introduction of CSIA has provided many extra dimensions to such studies. In the run up to the 2004 Olympics in Athens, SIBL took part in a study co-ordinated by European IOC laboratories, with veterinary and regulatory partners, to validate the use of 13C natural abundance to investigate the misuse of anabolic steroids (http://ec.europa.eu/research/growth/gcc/projects/antidoping-isotrace.html). Recently, we validated the use of 13C natural abundance in individual fatty acids to clearly demonstrate the provenance of wild and farmed sea fish.
Selected References
Fearon KCH, Hansell DT, Preston T, Plumb J.A., Davies J., Shapiro D., Shenkin A., Calman K.C. & Burns H.J.G. (1988) Influence of whole body protein turnover rate on resting energy expenditure in patients with cancer. Cancer Research, 48, 2590-2595.
Fearon KCH, McMillan DC, Preston T, Winstanley FP, Cruickshank AM and Shenkin A (1991) Elevated Circulating Interleukin-6 is Associated with an Acute-phase Response but Reduced Fixed Hepatic Protein Synthesis in Patients with Cancer, Annals of Surgery, 213 (1), 26-31
MacGregor BJ, Bruchert V, Fleishcher S & Amann R (2002) Isolation of small-sub-unit rRNA for stable isotopic characterization Environmental Microbiology 4(8): 451-464
Meselson M & Stahl FW (1958) The Replication of DNA in Escherichia Coli Proc Nat Ac Sci, 44: 671-682
Morrison, DJ, Cooper, K, Waldron, S, Slater, C, Weaver, LT & Preston, T, 2004, A streamlined approach to the analysis of volatile fatty acids and its application to the measurement of whole-body flux Rapid Communications In Mass Spectrometry 18(21): 2593-2600
Moses AWG, Slater C, Preston T, Barber MD, Fearon KCH, 2004, The reduced total energy expenditure and physical activity in cachectic patients with pancreatic cancer can be modulated by energy and protein dense oral supplements enriched with n-3 fatty acids. British Journal of Cancer 90 (5), 996-1002
Preston T & Slater C, (1994) Mass Spectrometric Analysis of Stable Isotope Labelled Amino Acid Tracers. Proceedings of the Nutrition Society, 53, 363-372
Radajewski, S., Ineson, P., Parekh, N.R., and Murrell, J.C. (2000) Stable-isotope probing as a tool in microbial ecology. Nature 403: 646–649
Selberg O, McMillan D C, Preston T, Carse H, Shenkin A and Burns H J G, (1990) Palmitate turnover and its response to glucose infusion in weight-losing cancer patients, Clinical Nutrition, 9, 150-156
Slater C, Preston T & Weaver LT (2006) Advantages of deuterium-labelled mixed triacylglycerol in studies of intraluminal fat digestion Rapid Communications in Mass Spectrometry 20 (2): 75-80
Slater C, Preston T, & Weaver LT (2006) Improving the specificity of the [C-13] mixed triacylglycerol breath test by estimating carbon dioxide production from heart rate European Journal Of Clinical Nutrition 60 (11): 1245-1252
Winter JW, Paterson S, Scobie G, Wirz A, Preston T & McColl KEL (2007) N-nitrosamine generation from ingested nitrate via nitric oxide in human subjects with and without gastro-esophageal reflux, Gastroenterology, in press
SUERC Isotope Biochemistry Laboratory is now at www.gla.ac.uk/suerc/isotopebiochemistry

