BMG Labtech Clariostar
The Clariostar is a multimode microplate reader equipped with two linear variable filter, (LVF), tuneable monochromators and filter selectors. One LVF monochromator is used for excitation and the other for emission. A linear variable dichroic mirror (LVDM) slide is used to efficiently separate excitation light from emission light. LVF monochromator has a linear variable long pass slide and short pass slide which form the rising edge and falling edge of a bandpass filter. Moving the long pass slide and short pass slide to their optimal position creates a filter with a distinct variable peak wavelength and bandwidth. This novel design ensures high transmission of emitted signals with excellent blockingof stray light. Both monochromators have tuneable wavelength selection, (range =320 to 850 nm), with bandwidth control, (range = 8 to 100 nm), for optimal excitation and detection of multifluorescence signals emitted from UV-, Visible-, or far Red fluorochrome dyes. The tuneable design allows weak dyes which have a poor quantum yield to be more efficiently excited by increasing the excitation bandwidth and vice versa bleedthrough of unwanted signals can be reduced by decreasing the bandwidth.
The system also has an ultra-fast spectrometer, (<1 sec/well), for performing UV/Vis absorbance measurement (220 to 1000 nm).
Features:
Endpoint or kinetic cell-based assays can be initiated with reagent injectors; and an integrated fluorophore library helps to set up assays
Detection modes:
Fluorescence intensity-including FRET, FI excitation or emission spectral scans
Luminescence (BRET)
Time-resolved FRET
UV/Vis absorbance spectra
Measurement modes:
Top/Bottom read
Endpoint/kinetic read
Sequential Multi Excitation measurements
Sequential Multi Emission measurements
Well scanning
Spectral scanning-Absorbance, Fluorescence, Luminescence
Microplate:
6-1536 plated
Light Source:
Xenon Flash lamp, Laser option not available
Detectors:
Low noise photomultiplier and CCD spectrometer
Z-adjustment:
Automatic focal height adjustment (0.1 mm resolution)
Reagent Injection:
2 built-in reagent injectors
Individual injection volumes for each well (3 to 350 µL)
Variable injection speed up to 420 µL / sec.
Up to four injection events per well
Reagent back flushing
Shaking:
Linear, orbital, and double-orbital with user-definable time and speed
Incubation:
+3°C above ambient up to 45°C or 65°C
Microscopy Live Cell Imaging and Plate Reader Multi-Fluorescence Technology
Professor Graeme’s Milligan’s Molecular Pharmacology group have a diverse range of multiflurescence equipment for exploring GPCR oligomerisation states and monitoring regulation of intracellular calcium. Located within the laboratory 253 there are two high resolution multi-fluorescence imaging microscopy systems for detecting fluorescence signals emitted from fluorescently tagged GPCRs in 2D or 3D over time. The laboratory is also equipped with five benchtop microplate readers, (BMG PHERAstar FS, BMG Omega Polarstar, MDS Flexstation II,Berthold Mithras LKB 940, Cellomics Arrayscan II,), for performing fast kinetic or end point cell based plate GPCR screening assays. GPCR assays routinely performed are time resolved FRET, (Tr-FRET), inter/intramolecular FRET, BRET, alphascreen and Fura-2 dual wavelength ratiometric monitoring of changes in intracellular Ca2+.
Microscopy Live Cell Multi-Fluorescence Imaging Systems:
Zeiss Axio Observer Z1 VivaTome Structured Illumination and Colibri LED Live Cell imaging system
This motorised XY and Z control invert microscope has temperature and CO2 control and has been set up to optimally record multi-fluorescent or FRET signals emitted from live cells in 2D or 3D over time. A VivaTome structured illumination spinning disk unit attached to the bottom port of the microscope allows high resolution confocal and wide-field images to be recorded simultaneously at high speed. Due to the XY and Z motorised capability, various image recording modes can be easily configured, (e.g. single, multichannel, z stack, time lapse and multifield imaging). The microscope has real time z focus drift correction, (Definite Focus), to overcome thermal induced Z focus drifting which can be problematic when recording long-term multifluorescence signals.
The microscope has also been fitted with a Colibri LED illumination excitation source for µsec high speed multifluorescence switching of excitation light to minimise photobleaching when performing multi-epifluorescence time lapse imaging or intramolecular FRET experiments where large image data sets are streamed directly on to the hard drive every 5-30 msecs with precise precision. Emitted fluorescence or FRET signals are sequentially or simultaneously detected using an EVOLVE 512 EMCCD camera coupled to a Dualview 2 image splitting device (Mag Biosystems) situated on the microscope’s left hand port. A programmable, high speed pressure driven solenoid valve superfusion system with triggered TLL control has also been set up to ensure accurate, rapid application of test ligands each time a high speed experiment is performed. The Colibri, Evolve 512 EMCCD camera and superfusion system are TTL controlled via a SVB1 trigger breakout box to synchronise high speed acquisition without any host computer interaction. A photograph of the Zeiss Axio Observer Z1 VivaTome/Colibri microscope imaging system is shown below.
Nikon Eclipse TE 2000-E Imaging Microscope
The NIKON ECLIPSE TE 2000-E invert microscope has motorised Z control and can be operated in multiple configurations which allows users to perform single or multiepifluorescence experiments at high speed. Excitation fluorescence over the wavelength range from 320 to 600 nm is rapidly generated from a Cairn optoscan monochromator and a 10 position emission filter wheel fitted to the LHS port of the microscope is used for optimal detection of blue, cyan, green, yellow and red monomeric fluorescent proteins or fluorochromes. Photobleaching is minimised by rapidly shuttering, (~ 1 msec), the output slit of the monochromator. Emitted fluorescence signals can be sequentially detected using a high resolution Cool Snap-HQ digital camera coupled to the emission filter wheel. Alternatively, fluorescence emissions can be detected simultaneously with no time delay using a Cool Snap HQ2 camera coupled to a Quadview 2 image splitting device (Mag Biosystems) situated on the microscope’s bottom port. The microscope has a built in linear z encoder for high precision acquisition of multifluorescence z stacks over time (4D). Rapid exchange of test ligands to cells grown on round glass coverslips is achieved by using an open cell perfusion chamber coupled to 2 solenoid valves via a 4-channel peristaltic pump. A picture of the Nikon microscope imaging system is illustrated below.
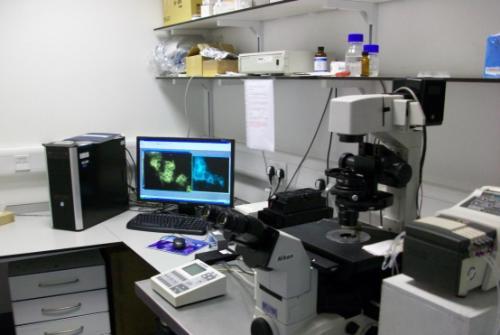
Plate Reader Multi-Fluorescence Technology Instruments:
BMG Labtech PHERAstar FS
PHERAstar FS instrument is our fastest reading multifluorescence plate reader and offers the highest spectral detector sensitivity. It is equipped with variable excitation sources, (i.e. high intensity Xenon Flash Lamp or solid state N2 UV laser for time resolved FRET and Alphascreen assays), and has two pairs of matched photomultiplier tubes (PMTs) with different spectral responses for optimal detection of BRET/luminescence, TR-FRET, HTRF, AlphaScreen, fluorescence polarisation and fluorescence intensity. This reader is also equipped with an ultra-fast UV/Visible spectrometer for quantifying absorbance, (220-1000 nm, maximal spectral resolution possible is 1 nm), and a real-time TR-FRET/AlphaScreen decay module allows TR-FRET or AlphaScreen assays to be maximally optimised. The system also has on-board reagent injectors for performing real time fast kinetic assays and precise temperature control allows live cell measurement at 37 degrees C. Sequential or simultaneous dual emission measurements can be also be quantified. Pherastar can read from 6 to 384 well plates and multiplate readings can be measured by inserting the plates into the stacker. Another useful feature is cell layer well scanning for multiple read measurements within a well. Well scanning can be performed on 6 to 1534 well plates. Plates can also be automatically shaken prior to reading and the instrument has the capability to read from the top or bottom of the microplate well . The photograph below shows the setup of the PHERAstar FS.

BMG Labtech Omega Polarstar
Omega Polarstar is equipped with a high energy Xenon Flash Lamp and has an 8 position excitation and emission filter wheel for sequential multi excitation and emission. System is equipped with two side window photomultiplier tubes, (spectral range 240-900 nm), for sequential or simultaneous dual emission quantification of fluorescence intensity, including FRET, fluorescence polarisation, Luminescence (flash and glow) - including BRET and Alphascreen. Bottom read absorbance measurements over the wavelength range 220-850 nm with selectable spectral resolution 1, 2 or 5 nm can be detected using the built in spectrometer detector. The system also has on-board reagent injectors for performing kinetic assays in real time and precise temperature control allows live cell measurement at 37 degrees C. The system is also equipped with a gas vent for CO2 incubation of living cells and a variety of plates can be read, (6 to 384 plates). Cell layer well scanning can be performed on 6 to 96 well format plates only and plates can also be automatically shaken prior to
Molecular Devices Flexstation II
Flexstation II has an integrated dual scanning monochromator for optimal multi sequential excitation and detection of assay multi-fluorescent probes. Fast kinetic assays are achieved by using an integrated 8 channel pipettor which can transfer multiple compound fluids rapidly from the reagents plate to the assay plate and data is acquired as soon as the compound fluid has been dispenced into the well. This property allows fast kinetic events, such as the mobilisation of calcium (Ca2+) from internal stores, extracellular Ca2+ influx into the cytosol via voltage operated Ca2+ channels or changes in membrane potential to be measured in real time without any time delay. The integrated dual scanning monochromators can perform excitation or emission spectral scans at 1 nm intervals over the excitation wavelength range from 250 to 850 nm and emission wavelength from 375-850 nm. This feature allows users to optimise their assays by determining the actual excitation and emission peak of the fluorescent dye they are working with. The instrument also has temperature control, automatic shaking and can perform top or bottom read measurements from 6-96 well microplates. Multiple read measurements within a well are also possible when the well scanning mode feature within the software is selected.
Cellomics Arrayscan II
Arrayscan II instrument is an automated high throughput multifluorescence microscopic imaging microplate reader for performing high throughput fluorescence cell-based assays in 96-well microplates. Acquired fluorescence images of the cells in each well are automatically analysed by proprietary bioapplication image processing algorithms which automatically convert images into numeric data that detects quantitate changes in intensity, cell size, shape and other properties in cells. Bioapplication assays available include: cytoplasm to nucleus translocation, receptor internalisation and trafficking, cell viability, mitotic index and multiparameter apoptosis. Cytoplasm to nucleus translocation assay quantifies translocation events by measuring the relative distribution of target fluorescence intensities between defined cytoplasm and nuclear regions of a cell, e.g. translocation of transcription factor nuclear factor kappa B, (NFB) from the cytoplasm to the nucleus. The receptor internalisation and trafficking application is designed for measuring and detecting internalised fluorescently tagged receptors that traffic to recycling endosomes. Green fluorescent ligands that bind to the receptor or GPCRs tagged at their C terminus with the green fluorescent protein eGFP in combination with the recycling endosomal marker transferrin-Alexa Fluor546 are ideal for performing this assay.
Evos FL Auto2 imaging station:
Our invert EVOS FL Auto2 imaging system is a bench top epifluorescence & transmitted light, (bright field and phase contrast) imaging station that can be automated to tile scan multi-well plates, culture flasks, petri-dishes and slides.

A dedicated monochrome camera, optimised for fluorescence detection, is used to record 16 bit depth multi-fluorescence channel images from live or fixed cell based samples. A separate color camera is also available for recording colorimetic images such as chromogenically stained immunohistochemistry tissue samples. An onstage incubator can also be fitted to perform a wide range of biological assays under physiological conditions. The system can be fitted with a wide range of LED/filter cubes to sequentially detect fluorescence emitted from dyes over the UV to IR range.
The 3 LED light cubes that we have fitted for sequentially acquiring single plane or z-stack multi-fluorescence channels over time are listed below:
DAPI: Ex = 357/44 nm. Em = 447/60 nm---------- -Cat no: AMEP4650
EGFP: Ex = 470/22 nm. Em = 510/42 nm-----------Cat no: AMEP4651
RFPI: Ex = 531/40 nm. Em = 593/40 nm------------Cat no: AMEP4652
Click on the following web link to see the full range of LED light cubes that are currently available to purchase and the dyes that can be detected using our fitted LED light cubes.
Stage x-y scanning speed:
Sequential 3 color multichannel fluorescence imaging of a 96 well plate takes < 5 minutes.
Fitted Objective Lens:
A diverse range of coverslip corrected objective lenses, (1.25 x to 100 x), can be purchased depending upon your requirements, e.g. long working distance, (WD), with low numerical aperture; (NA) & short WD with high NA. Lenses fitted are listed directly below:
Turret Position 1-cat no AMEP4699:
40 x Plan Fluorite Dry Air, NA = 0.75; WD = 0.89 mm, phase contrast NO.
Turret Position 2-cat no AMEP4622:
4 x Plan Fluorite Dry Air, NA = 0.13; WD = 19.7 mm; phase contrast NO.
Turret Position 3-cat no AMEP4734:
20 x Plan Apochromat Dry Air, NA = 0.75; WD = 0.77 mm; phase contrast NO.
Turret Position 4-cat no AMEP4682:
20 x Plan Fluorite Dry Air, NA = 0.4; WD = 3.1 mm; phase contrast YES..
Turret Position 5-cat no AMEP4700:
100 x Plan Fluorite Oil immersion, NA = 1.28; WD = 3.1 mm; phase contrast NO.
Other available objectives:
cat no AMEP4682:
2 x Plan Achromat Dry Air, NA = 0.06; WD = 5.1 mm; phase contrast NO.
cat no AMEP4681:
10 x Plan Fluorite Dry Air, NA = 0.25; WD = 9.2 mm; phase contrast YES.
cat no AMEP4753:
10 x Plan Apochromat Dry Air, NA = 0.4; WD = 3.27 mm; phase contrast NO
Stage plates and vessel holders:
We have a diverse range of stage plates and vessel holders which can be fitted to suit your sample needs and these are listed below.
Available stage plates
Master plate:
-cat no AMEP-VH035: master plate for the EVOS onstage incubator, accommodates environmental chamber. L = 185.3 mm, W = 135.3 mm.

Slides:
-cat no AMEP-VH021: holds 2 microscope slides or chamber slides with retention clip. L = 76.7 mm, W = 26.4 mm.
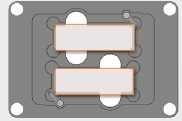
-cat no AMEP-VH067: holds 4 microscope slides with retention clip. L = 77 mm, W = 26 mm.
Well plates/well dishes:
-cat no AMEP-VH004: holds one Nunc 100 mm petri dish, D= 88.5 mm.

-cat no AMEP-VH022: holds one multiplate with retention clip for AMEPVH001 through to AMEPVH018. L = 128.2 mm, W = 86.2 mm.

Vessel holders:
Petri dish:
-cat no AMEP-VH029: onstage incubator, holds one 35 mm petri dish with retention clip, diameter = 41 mm.

Well plate:
-cat no AMEP-VH028: onstage incubator, holds one multi well plate with retention clip, L = 128.2 mm, W = 86.2 mm.

Features:
- Automated time lapse imaging including area scanning and multifield
- Z stack visualisation
- Movie avi creation
- Transmitted light: bright field/phase contrast/color and fluorescence


