Professor Cheryl Woolhead
- Professor of Molecular Biotechnology (Molecular Biosciences)
- Dean of Learning & Teaching (MVLS College Senior Management)
telephone:
01413305161
email:
Cheryl.Woolhead@glasgow.ac.uk
Level 4, Institute of MC&SB, Bower Building, Glasgow G12 8QQ
Research interests
Our group here at Glasgow is interested in the relationship between translating ribosome and nascent polypeptide. Currently this leads us into two main avenues of research.
Membrane protein folding and insertion
Inner membrane proteins make up 20-30% of the bacterial proteome. The major route of insertion for such proteins is via the Sec machinery. However certain membrane proteins are Sec independent and instead insert via an essential membrane protein called YidC. Despite the importance of YidC, the precise function and mechanism of this protein are difficult to define. In our lab we are using Fluorescence Resonance Energy Transfer (FRET) to monitor structural changes of an inner membrane protein during synthesis on the ribosome, targeting to the YidC pathway and integration into the membrane. This technique enables us to monitor the compaction of α-helices and the formation of tertiary structure, in order to elucidate the timing of folding events that occue between the ribosome exit tunnel and the adoption of the final functional conformation in the membrane. The findings are supported by in vitro insertion assays and crosslinking to the signal recognition (SRP), which provide further details of the role of SRP in YidC targeting.
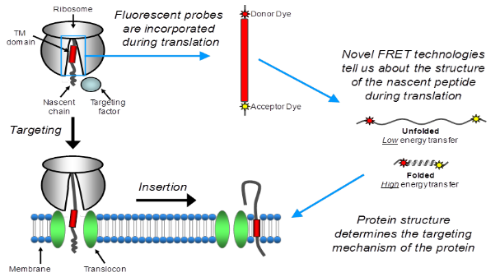
Figure 1. Structural analysis of membrane protein insertion
References:
Robinson, P. J., Findlay J. E. and Woolhead C. A. In Press (2012)
Woolhead, C. A., McCormick P. J. and Johnson A. E. Cell (2004)
Translation Arrest motifs
Although ribosomes are responsible for the synthesis of all cellular proteins it was initially believed that translating nascent chains would not interact with the exit tunnel during synthesis. However a small but increasing number of proteins have been identified which interact with the ribosome exit tunnel to induce translational arrest. Escherichia coli secretion monitor (SecM) is one such stalling peptide, SecM monitors the cell export activity through its own translocation to the periplasm and upregulates translation of SecA, an ATPase involved in the SecYEG translocation machinery, when translocation is reduced. How these peptides interact with the exit tunnel is not fully understood however two key features required for stalling include an essential peptide arrest motif and compaction of the nascent chain within the exit tunnel upon stalling. By investigating the SecM sequence found inside the ribosome tunnel and analysis of compaction of the nascent chain in the exit tunnel by structural and biochemical assays we are investigating the interactions between the nascent chain and the exit tunnel which contribute to translation arrest.
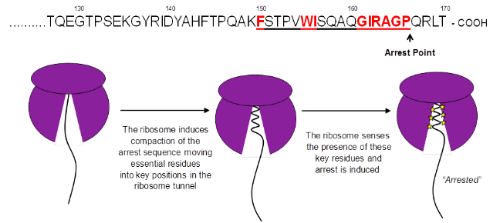
Figure 2. Nascent SecM induced translation arrest
References:
Woolhead, C. A. Johnson, A. E. and Bernstein H. D. Molecular Cell (2006)
Grants
Grants and Awards listed are those received whilst working with the University of Glasgow.
- Droplet-based Microfluidic Platform for Intracellular Ion Channel Drug Discovery
Engineering and Physical Sciences Research Council
2016 - 2019
- Folding and insertion of a bacterial inner membrane protein
Biotechnology and Biological Sciences Research Council
2010 - 2013
- Incorporation of fluorescently modified amino acids into proteins for structural studies
The Royal Society
2008 - 2009
Additional information
Invited International Presentations
- 2006: Rehovot, Israel - Kimmelman Center for Biomolecular Structure and Assembly, Weizmann Institute
- 2006: Maryland, USA - Institute for Physical Science and Technology University of Maryland, College Park, MD
- 2005: San Francisco, USA - American Society for Cell Biology 'Protein Folding & Quality Control' Symposium
- 2004: Hamburg,Germany - Spatio-Temporal Interactions of Cellular Signaling Molecules
- 2001: Acquafredda di Maratea, Italy - European Science Foundation: Protein Targeting
Professional Learned Society
- 2000 - present: Biochemical Society - Member
Research Fellowship
- 2005 - 2007: NIH NIDDK Scientific Directors Fellowship



