Professor Andrew Todd
- Professor of Neuroscience (Centre for Neuroscience)
telephone:
01413305868
email:
Andrew.Todd@glasgow.ac.uk
R157 Level 1, West Medical Building, Glasgow, G12 8qq
Research interests
Neurochemistry and Synaptic Connections in the Mammalian Spinal Cord
My main research interest is the organisation of neuronal circuits in the spinal dorsal horn that underlie the perception of pain and itch. We use immunocytochemistry and confocal microscopy combined with other techniques, such as neuronal tract-tracing, patch-clamp recording from spinal cord slices, and electron microscopy. I have collaborations with: Dr Marco Beato (UCL); Dr Toshi Yasaka (Saga University, Japan), Dr Sarah Ross (University of Pittsburgh, USA); Dr Peter Szucs and Prof Boris Safronov (University of Porto, Portugal); Dr Masahiko Watanabe (Hokkaido University, Japan). My work is funded by the Wellcome Trust and the BBSRC.
Organisation of neuronal circuits in the dorsal horn
Although the dorsal horn plays a major role in transmitting and modifying incoming sensory information, we know little about the nerve circuits responsible for these functions, largely due to the difficulty in defining functional populations of neurons.
Projection neurons
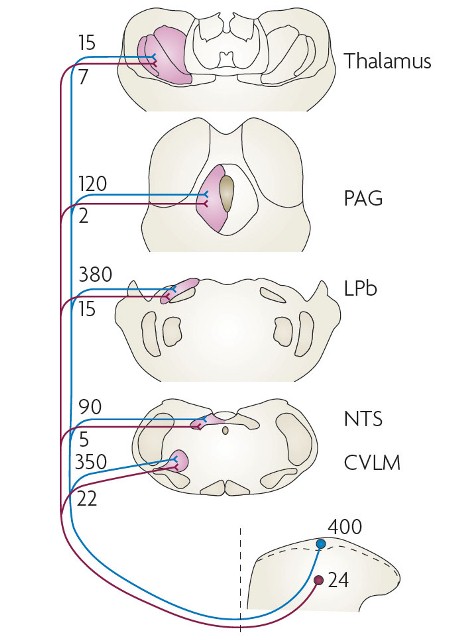
Projection neurons, with axons that travel directly to the brain, are concentrated in lamina I, and most of these express the neurokinin 1 receptor (NK1r). There are ~400 lamina I projection neurons per side in the rat L4 segment, corresponding to ~5% of all neurons in this lamina. Many project to more than one brain region. There are also NK1r-expressing projection neurons in lamina III, with similar supraspinal targets, and we have identified a population of giant lamina I projection cells that lack the NK1r.
Interneurons
Most neurons in laminae I-III have axons that remain in the spinal cord and are defined as interneurons. They can be divided into two main groups: excitatory and inhibitory. We have shown that among the inhibitory interneurons 4 populations can be defined by the expression of neuropeptide Y (NPY), galanin/dynorphin, neuronal nitric oxide synthase (nNOS) and parvalbumin. These account for over half the inhibitory interneurons in laminae I-II.
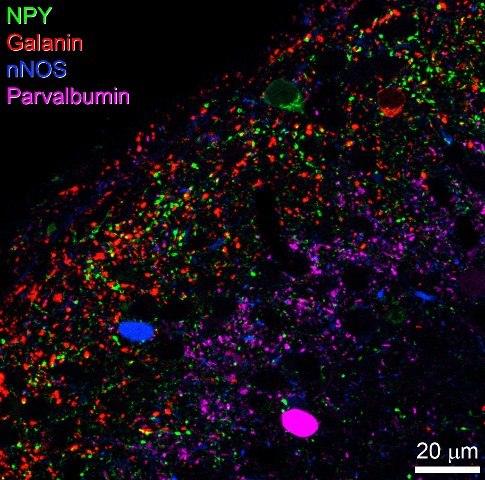
We have shown that they differ in their postsynaptic targets, e.g. NPY/GABA axons preferentially innervate NK1r+ lamina III projection cells, while nNOS/GABA axons innervate lamina I giant cells. Some of the neuronal circuits that have been identified in laminae I-III are summarised below.
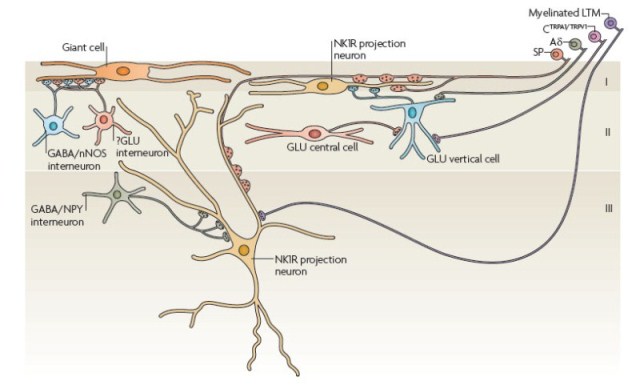
Changes in dorsal horn following nerve injury
Injury to peripheral nerves often leads to neuropathic pain, but the underlying mechanisms are poorly understood. There is evidence that changes in the dorsal horn contribute to certain types of neuropathic pain, and several possible mechanisms have been proposed. These include: (1) sprouting of Aβ low-threshold mechanoreceptive afferents into the superficial laminae, where they would have access to nociceptive-specific projection neurons; (2) a phenotypic switch, such that these afferents start to synthesise substance P, which could activate NK1rs on nociceptive projection neurons; (3) loss of inhibitory interneurons from the superficial laminae.
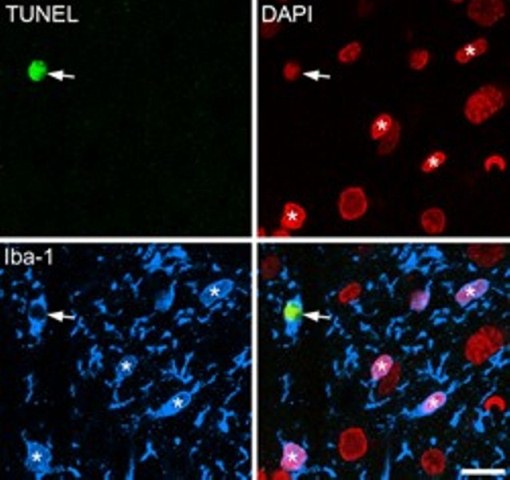
We have provided evidence against each of these suggestions. We found that both before and after nerve injury, the central projections of Aβ afferents extend into, but not beyond the inner half of lamina II. We also showed that the central terminals of these afferents did not express or release substance P after nerve injury. We found that in rat models of neuropathic pain, there was no change in the total number of neurons in laminae I-III, nor in the proportion that were GABA-immunoreactive. There is known to be apoptotic cell death in the dorsal horn in these models, as shown by staining with the TUNEL (Terminal deoxynucleotidyl transferase dUTP nick end labeling) method. However, we showed that this was restricted to microglia (labelled with Iba1 antibody), and that TUNEL positive cells were not neurons.
Interneurons in the ventral horn
Although most of our work is on the dorsal horn, we have also identified two important populations of ventral horn interneurons: those giving rise to the "P" and "C" boutons that are associated with motoneurons.
P boutons are GABAergic terminals that form axoaxonic synapses on Ia muscle spindle afferents and regulate proprioceptive input to motoneurons. We showed that P boutons were unique in expressing the 65kDa molecular weight form of glutamic acid decarboxylase (GAD65) This enabled us to identify their cells of origin, which were in the deep medial dorsal horn, and showed that P boutons are likely to represent the only output for these cells.
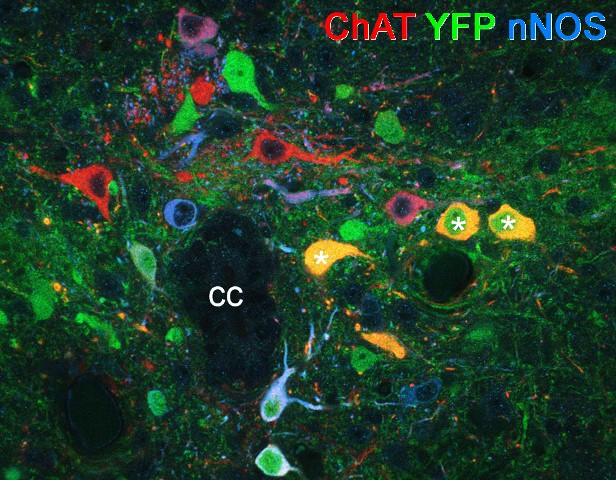
The C boutons are cholinergic axons that regulate motoneuronexcitability through m2 muscarinic receptors. In collaboration with Drs Rob Brownstone and Gareth Miles (Dalhousie University, Canada) we used a neurochemical/developmental approach to demonstrate that these boutons originate from a group of cholinergic cells located lateral to the central canal (cc) – the so-called "medial partition cells". This was based on the finding that the C boutons, which express choline acetyltransferase (ChAT), lack neuronal nitric oxide synthase (nNOS) but transiently express the transcription factor Dbx1, which was captured with yellow fluorescent protein (YFP).
Publications
2023
Quillet, R., Gutierrez-Mecinas, M. , Polgár, E., Dickie, A. C., Boyle, K. A., Watanabe, M. and Todd, A. J. (2023) Synaptic circuits involving gastrin-releasing peptide receptor-expressing neurons in the dorsal horn of the mouse spinal cord. Frontiers in Molecular Neuroscience, 16, 1294994. (doi: 10.3389/fnmol.2023.1294994) (PMID:38143564) (PMCID:PMC10742631)
Boyle, K. A. et al. (2023) Neuropeptide Y-expressing dorsal horn inhibitory interneurons gate spinal pain and itch signalling. eLife, 12, RP86633. (doi: 10.7554/eLife.86633) (PMID:37490401) (PMCID:PMC10392120)
Davis, O. C. et al. (2023) Calretinin-expressing islet cells are a source of pre- and post-synaptic inhibition of non-peptidergic nociceptor input to the mouse spinal cord. Scientific Reports, 13, 11561. (doi: 10.1038/s41598-023-38605-9) (PMID:37464016) (PMCID:PMC10354228)
Quillet, R., Dickie, A. C., Polgár, E., Gutierrez-Mecinas, M. , Bell, A. M. , Goffin, L., Watanabe, M. and Todd, A. J. (2023) Characterisation of NPFF-expressing neurons in the superficial dorsal horn of the mouse spinal cord. Scientific Reports, 13, 5891. (doi: 10.1038/s41598-023-32720-3) (PMID:37041197) (PMCID:PMC10090074)
Gutierrez-Mecinas, M. , Kókai, É., Polgár, E., Quillet, R., Titterton, H. F., Weir, G. A. , Watanabe, M. and Todd, A. J. (2023) Antibodies against the gastrin-releasing peptide precursor pro-gastrin-releasing peptide reveal its expression in the mouse spinal dorsal horn. Neuroscience, 510, pp. 60-71. (doi: 10.1016/j.neuroscience.2022.12.023) (PMID:36581131)
Ronzano, R. et al. (2023) Spinal premotor interneurons controlling antagonistic muscles are spatially intermingled. eLife, 11, e81976. (doi: 10.7554/elife.81976) (PMID:36512397) (PMCID:PMC9844990)
Polgár, E. et al. (2023) Grpr expression defines a population of superficial dorsal horn vertical cells that have a role in both itch and pain. Pain, 164(1), pp. 149-170. (doi: 10.1097/j.pain.0000000000002677) (PMID:35543635) (PMCID:PMC9756441)
2022
Trendafilova, T. et al. (2022) Sodium-calcium exchanger-3 regulates pain "wind-up": from human psychophysics to spinal mechanisms. Neuron, 110(16), 2571-2587.e13. (doi: 10.1016/j.neuron.2022.05.017) (PMID:35705078) (PMCID:PMC7613464)
Hughes, D. I. and Todd, A. J. (2022) Visualizing synaptic connectivity using confocal and electron microscopy: neuroanatomical approaches to define spinal circuits. In: Contemporary Approaches to the Study of Pain. Series: Neuromethods, 178. Humana: New York, pp. 181-201. ISBN 9781071620380 (doi: 10.1007/978-1-0716-2039-7_11)
El Khoueiry, C., Alba-Delgado, C., Antri, M., Gutierrez-Mecinas, M. , Todd, A. J. , Artola, A. and Dallel, R. (2022) GABAA and glycine receptor-mediated inhibitory synaptic transmission onto adult rat lamina IIi PKCγ-interneurons: pharmacological but not anatomical specialization. Cells, 11(8), 1356. (doi: 10.3390/cells11081356) (PMID:35456035) (PMCID:PMC9033052)
Todd, A. J. (2022) An historical perspective: the second order neuron in the pain pathway. Frontiers in Pain Research, 3, 845211. (doi: 10.3389/fpain.2022.845211) (PMID:35295811) (PMCID:PMC8915774)
Kókai, É., Alsulaimain, W. A.A., Dickie, A. C., Bell, A. M. , Goffin, L., Watanabe, M., Gutierrez-Mecinas, M. and Todd, A. J. (2022) Characterisation of deep dorsal horn projection neurons in the spinal cord of the Phox2a::Cre mouse line. Molecular Pain, 18, p. 17448069221119614. (doi: 10.1177/17448069221119614) (PMID:36000342) (PMCID:PMC9445510)
2021
Alsulaiman, W. A.A. et al. (2021) Characterisation of lamina I anterolateral system neurons that express Cre in a Phox2a-Cre mouse line. Scientific Reports, 11, 17912. (doi: 10.1038/s41598-021-97105-w) (PMID:34504158) (PMCID:PMC8429737)
Chisholm, K. I., Lo Re, L., Polgár, E., Gutierrez-Mecinas, M. , Todd, A. J. and McMahon, S. B. (2021) Encoding of cutaneous stimuli by lamina I projection neurons. Pain, 162(9), pp. 2405-2417. (doi: 10.1097/j.pain.0000000000002226) (PMID:33769365) (PMCID:PMC8374708)
2020
Polgár, E., Bell, A. M. , Gutierrez-Mecinas, M. , Dickie, A. C., Akar, O., Costreie, M., Watanabe, M. and Todd, A. J. (2020) Substance P-expressing neurons in the superficial dorsal horn of the mouse spinal cord: insights into their functions and their roles in synaptic circuits. Neuroscience, 450, pp. 113-125. (doi: 10.1016/j.neuroscience.2020.06.038) (PMID:32634530)
Bell, A. M. , Gutierrez-Mecinas, M. , Stevenson, A., Casas-Benito, A., Wildner, H., West, S. J., Watanabe, M. and Todd, A. J. (2020) Expression of green fluorescent protein defines a specific population of lamina II excitatory interneurons in the GRP::eGFP mouse. Scientific Reports, 10, 13176. (doi: 10.1038/s41598-020-69711-7) (PMID:32764601) (PMCID:PMC7411045)
Hughes, D. I. and Todd, A. (2020) Central nervous system targets: inhibitory interneurons in the spinal cord. Neurotherapeutics, 17(3), pp. 874-885. (doi: 10.1007/s13311-020-00936-0) (PMID:33029722)
Tran, M., Braz, J. M., Hamel, K., Kuhn, J., Todd, A. J. and Basbaum, A. I. (2020) Ablation of spinal cord estrogen receptor α-expressing interneurons reduces chemically induced modalities of pain and itch. Journal of Comparative Neurology, 528(10), pp. 1629-1643. (doi: 10.1002/cne.24847) (PMID:31872868) (PMCID:PMC7317200)
Peirs, C., Dallel, R. and Todd, A. J. (2020) Recent advances in our understanding of the organization of dorsal horn neuron populations and their contribution to cutaneous mechanical allodynia. Journal of Neural Transmission, 127, pp. 505-525. (doi: 10.1007/s00702-020-02159-1) (PMID:32239353) (PMCID:PMC7148279)
Bell, A. M. , Polgár, E., Gutierrez-Mecinas, M. , Boyle, K. A. and Todd, A. J. (2020) Functional populations among interneurons in the dorsal horn. In: Fritzsch, B. (ed.) The Senses: A Comprehensive Reference. Second Edition. Academic Press: Cambridge, MA, pp. 207-219. ISBN 9780128054093 (doi: 10.1016/B978-0-12-809324-5.24171-0)
2019
Gutierrez-Mecinas, M. , Bell, A. M. , Shepherd, F., Polgár, E., Watanabe, M., Furuta, T. and Todd, A. J. (2019) Expression of cholecystokinin by neurons in mouse spinal dorsal horn. Journal of Comparative Neurology, 527(11), pp. 1857-1871. (doi: 10.1002/cne.24657) (PMID:30734936) (PMCID:PMC6563475)
Dickie, A. C. et al. (2019) Morphological and functional properties distinguish the substance P and gastrin-releasing peptide subsets of excitatory interneuron in the spinal cord dorsal horn. Pain, 160(2), pp. 442-462. (doi: 10.1097/j.pain.0000000000001406) (PMID:30247267) (PMCID:PMC6330098)
Gutierrez-Mecinas, M. , Davis, O., Polgár, E., Shahzad, M., Navarro-Batista, K., Furuta, T., Watanabe, M., Hughes, D. I. and Todd, A. J. (2019) Expression of calretinin among different neurochemical classes of interneuron in the superficial dorsal horn of the mouse spinal cord. Neuroscience, 398, pp. 171-181. (doi: 10.1016/j.neuroscience.2018.12.009) (PMID:30553791) (PMCID:PMC6347472)
Gutierrez-Mecinas, M. , Bell, A. , Polgár, E., Watanabe, M. and Todd, A. J. (2019) Expression of neuropeptide FF defines a population of excitatory interneurons in the superficial dorsal horn of the mouse spinal cord that respond to noxious and pruritic stimuli. Neuroscience, 416, pp. 281-293. (doi: 10.1016/j.neuroscience.2019.08.013) (PMID:31421202) (PMCID:PMC6839401)
2018
Gutierrez-Mecinas, M. , Polgár, E., Bell, A. M. , Hérau, M. and Todd, A. J. (2018) Substance P-expressing excitatory interneurons in the mouse superficial dorsal horn provide a propriospinal input to the lateral spinal nucleus. Brain Structure and Function, 223(5), pp. 2377-2392. (doi: 10.1007/s00429-018-1629-x) (PMID:29497838) (PMCID:PMC5968060)
Huang, J. et al. (2018) Author correction: circuit dissection of the role of somatostatin in itch and pain. Nature Neuroscience, 21(6), p. 894. (doi: 10.1038/s41593-018-0149-6) (PMID:29674654)
Strnadel, J. et al. (2018) Survival of syngeneic and allogeneic iPSC–derived neural precursors after spinal grafting in minipigs. Science Translational Medicine, 10(440), eaam6651. (doi: 10.1126/scitranslmed.aam6651) (PMID:29743351)
Huang, J. et al. (2018) Circuit dissection of the role of somatostatin in itch and pain. Nature Neuroscience, 21(5), pp. 707-716. (doi: 10.1038/s41593-018-0119-z) (PMID:29556030) (PMCID:PMC5923877)
Dawes, J. M. et al. (2018) Immune or genetic-mediated disruption of CASPR2 causes pain hypersensitivity due to enhanced primary afferent excitability. Neuron, 97(4), 806-822.e10. (doi: 10.1016/j.neuron.2018.01.033) (PMID:29429934)
2017
Boyle, K. A., Gutierrez-Mecinas, M. , Polgár, E., Mooney, N., O'Connor, E., Furuta, T., Watanabe, M. and Todd, A. J. (2017) A quantitative study of neurochemically-defined populations of inhibitory interneurons in the superficial dorsal horn of the mouse spinal cord. Neuroscience, 363, pp. 120-133. (doi: 10.1016/j.neuroscience.2017.08.044) (PMID:28860091) (PMCID:PMC5648048)
Gutierrez-Mecinas, M. , Bell, A. M. , Marin, A., Taylor, R., Boyle, K. A., Furuta, T., Watanabe, M., Polgár, E. and Todd, A. J. (2017) Preprotachykinin A (PPTA) is expressed by a distinct population of excitatory neurons in the mouse superficial spinal dorsal horn including cells that respond to noxious and pruritic stimuli. Pain, 158(3), pp. 440-456. (doi: 10.1097/j.pain.0000000000000778) (PMID:27902570) (PMCID:PMC5302415)
Todd, A. J. (2017) Identifying functional populations among the interneurons in laminae I-III of the spinal dorsal horn. Molecular Pain, 13, pp. 1-19. (doi: 10.1177/1744806917693003) (PMID:28326935) (PMCID:PMC5315367)
2016
Bell, A. M. , Gutierrez-Mecinas, M. , Polgár, E. and Todd, A. J. (2016) Spinal neurons that contain gastrin-releasing peptide seldom express Fos or phosphorylate extracellular signal-regulated kinases in response to intradermal chloroquine. Molecular Pain, 12, pp. 1-9. (doi: 10.1177/1744806916649602) (PMID:27270268) (PMCID:PMC4937990)
Gutierrez-Mecinas, M. , Furuta, T., Watanabe, M. and Todd, A. J. (2016) A quantitative study of neurochemically-defined excitatory interneuron populations in laminae I-III of the mouse spinal cord. Molecular Pain, 12, pp. 1-18. (doi: 10.1177/1744806916629065) (PMID:27030714) (PMCID:PMC4946630)
Gutierrez-Mecinas, M. , Kuehn, E. D., Abraira, V. E., Polgár, E., Watanabe, M. and Todd, A. J. (2016) Immunostaining for Homer reveals the majority of excitatory synapses in laminae I-III of the mouse spinal dorsal horn. Neuroscience, 329, pp. 171-181. (doi: 10.1016/j.neuroscience.2016.05.009) (PMID:27185486) (PMCID:PMC4915440)
Chiang, M.C., Hachisuka, J. , Todd, A.J. and Ross, S.E. (2016) Erratum. Pain, 157(4), p. 992. (doi: 10.1097/j.pain.0000000000000552) (PMID:26982518)
Chiang, M. C., Hachisuka, J. , Todd, A. J. and Ross, S. E. (2016) Insight into B5-I spinal interneurons and their role in the inhibition of itch and pain. Pain, 157(3), pp. 544-545. (doi: 10.1097/j.pain.0000000000000474) (PMID:26716996)
Iwagaki, N., Ganley, R. P., Dickie, A., Polgár, E., Hughes, D. I. , Del Rio, P., Revina, Y., Watanabe, M., Todd, A. J. and Riddell, J. S. (2016) A combined electrophysiological and morphological study of NPY-expressing inhibitory interneurons in the spinal dorsal horn of the mouse. Pain, 157(3), pp. 598-612. (doi: 10.1097/j.pain.0000000000000407) (PMID:26882346) (PMCID:PMC4751741)
Todd, A. J. (2016) Anatomy of pain pathways. In: Battaglia, A. A. (ed.) An Introduction to Pain and its Relation to Nervous System Disorders. Wiley Blackwell, pp. 11-33. ISBN 9781118455913 (doi: 10.1002/9781118455968.ch1)
2015
Ganley, R. P. et al. (2015) Inhibitory interneurons that express GFP in the PrP-GFP mouse spinal cord are morphologically heterogeneous, innervated by several classes of primary afferent and include lamina I projection neurons among their postsynaptic targets. Journal of Neuroscience, 35(19), pp. 7626-7642. (doi: 10.1523/JNEUROSCI.0406-15.2015) (PMID:25972186) (PMCID:PMC4429159)
Cameron, D., Gutierrez-Mecinas, M. , Gomez-Lima, M., Watanabe, M., Polgár, E. and Todd, A. J. (2015) The organisation of spinoparabrachial neurons in the mouse. Pain, 156(10), pp. 2061-2071. (doi: 10.1097/j.pain.0000000000000270) (PMID:26101837) (PMCID:PMC4770364)
Todd, A. J. (2015) Plasticity of inhibition in the spinal cord. In: Schaible, H.-G. (ed.) Pain Control. Series: Handbook of experimental pharmacology. Springer, pp. 171-190. ISBN 9783662464496 (doi: 10.1007/978-3-662-46450-2_9)
2014
Baseer, N., Al-Baloushi, A. S., Watanabe, M., Shehab, S. A.S. and Todd, A. J. (2014) Selective innervation of NK1 receptor–lacking lamina I spinoparabrachial neurons by presumed nonpeptidergic Aδ nociceptors in the rat. Pain, 155(11), pp. 2291-2300. (doi: 10.1016/j.pain.2014.08.023) (PMID:25168670) (PMCID:PMC4247378)
Bhumbra, G. S., Bannatyne, B. A., Watanabe, M., Todd, A. J. , Maxwell, D. J. and Beato, M. (2014) The recurrent case for the Renshaw cell. Journal of Neuroscience, 34(38), pp. 12919-12932. (doi: 10.1523/JNEUROSCI.0199-14.2014) (PMID:25232126) (PMCID:PMC4166169)
Kardon, A. P. et al. (2014) Dynorphin acts as a neuromodulator to inhibit itch in the dorsal horn of the spinal cord. Neuron, 82(3), pp. 573-586. (doi: 10.1016/j.neuron.2014.02.046) (PMID:24726382) (PMCID:PMC4022838)
Ross, S. E., Hachisuka, J. and Todd, A. J. (2014) Spinal microcircuits and the regulation of itch. In: Carstens, E. and Akiyama, T. (eds.) Itch: Mechanisms and Treatment. CRC Press Taylor & Francis Group. ISBN 9781466505438
Gutierrez-Mecinas, M. , Watanabe, M. and Todd, A. J. (2014) Expression of gastrin-releasing peptide by excitatory interneurons in the mouse superficial dorsal horn. Molecular Pain, 10, 79. (doi: 10.1186/1744-8069-10-79) (PMID:25496164) (PMCID:PMC4320531)
Yasaka, T., Tiong, S. Y.X., Polgár, E., Watanabe, M., Kumamoto, E., Riddell, J. S. and Todd, A. J. (2014) A putative relay circuit providing low-threshold mechanoreceptive input to lamina I projection neurons via vertical cells in lamina II of the rat dorsal horn. Molecular Pain, 10(1), p. 3. (doi: 10.1186/1744-8069-10-3)
2013
Polgár, E., Sardella, T.C.P., Tiong, S.Y.X., Locke, S., Watanabe, M. and Todd, A.J. (2013) Functional differences between neurochemically defined populations of inhibitory interneurons in the rat spinal dorsal horn. Pain, 154(12), pp. 2606-2615. (doi: 10.1016/j.pain.2013.05.001)
Szucs, P., Luz, L.L., Pinho, R., Aguiar, P., Antal, Z., Tiong, S.Y.X., Todd, A.J. and Safronov, B.V. (2013) Axon diversity of lamina I local-circuit neurons in the lumbar spinal cord. Journal of Comparative Neurology, 521(12), pp. 2719-2741. (doi: 10.1002/cne.23311)
Iwagaki, N., Garzillo, F., Polgár, E., Riddell, J.S. and Todd, A.J. (2013) Neurochemical characterisation of lamina II inhibitory interneurons that express GFP in the PrP-GFP mouse. Molecular Pain, 9(56), (doi: 10.1186/1744-8069-9-56)
Polgár, E., Durrieux, C., Hughes, D.I. and Todd, A.J. (2013) A quantitative study of inhibitory interneurons in laminae I-III of the mouse spinal dorsal horn. PLoS ONE, 8(10), e78309. (doi: 10.1371/journal.pone.0078309) (PMID:24205193) (PMCID:PMC3808353)
2012
Baseer, N., Watanabe, M., Furuta, T., Kaneko, T. and Todd, A.J. (2012) Projection neurons in lamina III of the rat spinal cord are selectively innervated by local dynorphin-containing excitatory neurons. Journal of Neuroscience, 32(34), pp. 11854-11863. (doi: 10.1523/JNEUROSCI.2707-12.2012)
Tappe-Theodor, A. et al. (2012) Gαq/11 signaling tonically modulates nociceptor function and contributes to activity-dependent sensitization. Pain, 153(1), pp. 184-196. (doi: 10.1016/j.pain.2011.10.014)
Baseer, N., Polgar, E., Watanabe, M., Furuta, T., Kaneko, T. and Todd, A. (2012) Projection neurons in Lamina III of the rat spinal cord are selectively innervated by local dynorphin-containing excitatory neurons. Journal of Neuroscience, 32(34), pp. 11854-11863. (doi: 10.1523/JNEUROSCI.2707-12.2012)
Luo, C. et al. (2012) Presynaptically localized cyclic GMP-dependent protein kinase 1 is a key determinant of spinal synaptic potentiation and pain hypersensitivity. PLoS Biology, 10(3), e1001283. (doi: 10.1371/journal.pbio.1001283)
Todd, A.J. (2012) How to recognise collateral damage in partial nerve injury models of neuropathic pain. Pain, 153(1), pp. 11-12. (doi: 10.1016/j.pain.2011.10.031)
2011
Tiong, S. Y.X., Polgár, E., van Kralingen, J. C., Watanabe, M. and Todd, A. J. (2011) Galanin-immunoreactivity identifies a distinct population of inhibitory interneurons in laminae I-III of the rat spinal cord. Molecular Pain, 7, 36. (doi: 10.1186/1744-8069-7-36) (PMID:21569622) (PMCID:PMC3118366)
Polgár, E., Sardella, T.C.P., Watanabe, M. and Todd, A.J. (2011) Quantitative study of NPY-expressing GABAergic neurons and axons in rat spinal dorsal horn. Journal of Comparative Neurology, 519(6), pp. 1007-1023. (doi: 10.1002/cne.22570)
Saywell, S.A., Ford, T.W., Meehan, C.F., Todd, A.J. and Kirkwood, P.A. (2011) Electrophysiological and morphological characterization of propriospinal interneurons in the thoracic spinal cord. Journal of Neurophysiology, 105(2), pp. 806-826. (doi: 10.1152/jn.00738.2010)
Gangadharan, V. et al. (2011) Peripheral calcium-permeable AMPA receptors regulate chronic inflammatory pain in mice. Journal of Clinical Investigation, 121(4), pp. 1608-1623. (doi: 10.1172/JCI44911)
Sardella, T.C.P., Polgár, E., Garzillo, F., Furuta, T., Kaneko, T., Watanabe, M. and Todd, A.J. (2011) Dynorphin is expressed primarily by GABAergic neurons that contain galanin in the rat dorsal horn. Molecular Pain, 7(1), p. 76. (doi: 10.1186/1744-8069-7-76)
Sardella, T.C.P., Polgár, E., Watanabe, M. and Todd, A.J. (2011) A quantitative study of neuronal nitric oxide synthase expression in laminae I–III of the rat spinal dorsal horn. Neuroscience, 192, pp. 708-720. (doi: 10.1016/j.neuroscience.2011.07.011)
2010
Todd, A.J. (2010) Neuronal circuitry for pain processing in the dorsal horn. Nature Reviews Neuroscience, 11(12), pp. 823-836. (doi: 10.1038/nrn2947)
Yasaka, T., Tiong, S.Y.X., Hughes, D.I. , Riddell, J.S. and Todd, A.J. (2010) Populations of inhibitory and excitatory interneurons in lamina II of the adult rat spinal dorsal horn revealed by a combined electrophysiological and anatomical approach. Pain, 151(2), pp. 475-488. (doi: 10.1016/j.pain.2010.08.008)
Polgár, E., Wright, L. L. and Todd, A. J. (2010) A quantitative study of brainstem projections from lamina I neurons in the cervical and lumbar enlargement of the rat. Brain Research, 1308, pp. 58-67. (doi: 10.1016/j.brainres.2009.10.041)
Polgár, E., Al Ghamdi, K.S. and Todd, A.J. (2010) Two populations of neurokinin 1 receptor-expressing projection neurons in lamina I of the rat spinal cord that differ in AMPA receptor subunit composition and density of excitatory synaptic input. Neuroscience, 167(4), pp. 1192-1204. (doi: 10.1016/j.neuroscience.2010.03.028)
2009
Al Ghamdi, K.S., Polgár, E. and Todd, A. (2009) Soma size distinguishes projection neurons from neurokinin 1 receptor-expressing interneurons in lamina I of the rat lumbar spinal dorsal horn. Neuroscience, 164(4), pp. 1794-1804. (doi: 10.1016/j.neuroscience.2009.09.071)
Yasaka, T., Hughes, D.I. , Polgár, E., Nagy, G.G., Watanabe, M., Riddell, J.S. and Todd, A.J. (2009) Evidence against AMPA receptor-lacking glutamatergic synapses in the superficial dorsal horn of the rat spinal cord. Journal of Neuroscience, 29(42), pp. 13401-13409. (doi: 10.1523/JNEUROSCI.2628-09.2009)
Todd, A. J. , Polgár, E., Watt, C., Bailey, M. E.S. and Watanabe, M. (2009) Neurokinin 1 receptor-expressing projection neurons in laminae III and IV of the rat spinal cord have synaptic AMPA receptors that contain GluR2, GluR3 and GluR4 subunits. European Journal of Neuroscience, 29(4), pp. 718-726. (doi: 10.1111/j.1460-9568.2009.06633.x) (PMID:19200070) (PMCID:PMC2695158)
Al-Khater, K.M. and Todd, A.J. (2009) Collateral projections of neurons in laminae I, III, and IV of rat spinal cord to thalamus, periaqueductal gray matter, and lateral parabrachial area. Journal of Comparative Neurology, 515(6), pp. 629-646. (doi: 10.1002/cne.22081)
2008
Polgar, E., Al-Khater, K.M., Shehab, S., Watanabe, M. and Todd, A.J. (2008) Large projection neurons in lamina I of the rat spinal cord that lack the neurokinin 1 receptor are densely innervated by VGLUT2-containing axons and possess GluR4-containing AMPA receptors. Journal of Neuroscience, 28(49), pp. 13150-13160. (doi: 10.1523/JNEUROSCI.4053-08.2008)
Polgár, E. and Todd, A. (2008) Tactile allodynia can occur in the spared nerve injury model in the rat without selective loss of GABA or GABAA receptors from synapses in laminae I–II of the ipsilateral spinal dorsal horn. Neuroscience, 156(1), pp. 193-202. (doi: 10.1016/j.neuroscience.2008.07.009)
Polgár, E., Watanabe, M., Hartmann, B., Grant, S. and Todd, A.J. (2008) Expression of AMPA receptor subunits at synapses in laminae I-III of the rodent spinal dorsal horn. Molecular Pain, 4(5), pp. 1-14. (doi: 10.1186/1744-8069-4-5)
Al-Khater, K.M., Kerr, R. and Todd, A.J. (2008) A quantitative study of spinothalamic neurons in laminae I, III, and IV in lumbar and cervical segments of the rat spinal cord. Journal of Comparative Neurology, 511(1), pp. 1-18. (doi: 10.1002/cne.21811)
2007
Hughes, D.I. , Scott, D.T., Riddell, J.S. and Todd, A.J. (2007) Up-regulation of substance P in low-threshold myelinated afferents is not required for tactile allodynia in the chronic constriction injury and spinal nerve ligation models. Journal of Neuroscience, 27(8), pp. 2035-2044. (doi: 10.1523/JNEUROSCI.5401-06.2007)
Marvizón, J.C.G., Pérez, O.A., Song, B., Chen, W., Bunnett, N.W., Grady, E.F. and Todd, A.J. (2007) Calcitonin receptor-like receptor and receptor activity modifying protein 1 in the rat dorsal horn: localization in glutamatergic presynaptic terminals containing opioids and adrenergic α2C receptors. Journal of Neuroscience, 148(1), pp. 250-265. (doi: 10.1016/j.neuroscience.2007.05.036)
Miles, G.B., Hartley, R., Todd, A.J. and Brownstone, R.M. (2007) Spinal cholinergic interneurons regulate the excitability of motoneurons during locomotion. Proceedings of the National Academy of Sciences of the United States of America, 104(7), pp. 2448-2453.
Polgár, E., Campbell, A.D., MacIntyre, L., Watanabe, M. and Todd, A.J. (2007) Phosphorylation of ERK in neurokinin 1 receptor-expressing neurons in laminae III and IV of the rat spinal dorsal horn following noxious stimulation. Molecular Pain, 3(4),
Hughes, D.I. , Scott, D.T., Riddell, J.S. and Todd, A.J. (2007) Upregulation of substance P in low-threshold myelinated afferents is not required for tactile allodynia in the chronic constriction injury and spinal nerve ligation models. Journal of Neuroscience, 27(8), pp. 2035-2044. (doi: 10.1523/JNEUROSCI.5401-06.2007)
Marvizon, J., Perez, O., Song, B., Chen, W., Bunnett, N., Grady, E. and Todd, A. (2007) Calcitonin receptor-like and receptor activity modifying protein 1 in the rat dorsal horn localization in glutamatergic presynaptic terminal containing opioids and adrenergic alpha(2c) receptor. Neuroscience, 148, pp. 250-265. (doi: 10.1016/j.neuroscience.2007.05.036)
Polgar, E., Thomson, S., Maxwell, D., Al-Khater, K. and Todd, A. (2007) A population of large neurons in laminae III and IV of the rat spinal cord that have long dorsal dendrites and lack the neurokinin 1 receptor. European Journal of Neuroscience, 26, pp. 1587-1598. (doi: 10.1111/j.1460-9568.2007.05793.x)
2006
Polgar, E., Furuta, T., Kaneko, T. and Todd, A. (2006) Characterization of neurons that express preprotachykinin B in the dorsal horn of the rat spinal cord. Neuroscience, 139, pp. 687-697. (doi: 10.1016/j.neuroscience.2005.12.021)
Todd, A. (2006) Erratum to 'Characterization of neurons that express preprotachykinin B in the dorsal horn of the rat spinal cord': [Neuroscience 139 (2006) 687-697]. Neuroscience, 141(4), pp. 2195-2197. (doi: 10.1016/j.neuroscience.2005.12.0)
2005
Shehab, S., McGonigle, D., Hughes, D.I. , Todd, A.J. and Redgrave, P. (2005) Anatomical evidence for an anticonvulsant relay in the rat ventromedial medulla. European Journal of Neuroscience, 22(6), pp. 1431-1444. (doi: 10.1111/j.1460-9568.2005.04326.x)
Polgar, E., Hughes, D.I. , Arham, A.Z. and Todd, A.J. (2005) Loss of neurons from laminas I-III of the spinal dorsal horn is not required for development of tactile allodynia in the spared nerve injury model of neuropathic pain. Journal of Neuroscience, 25(28), pp. 6658-6666. (doi: 10.1523/JNEUROSCI.1490-05.2005)
Wilson, J. W., Hartley, R., Maxwell, D. J. , Todd, A. J. , Lieberam, I., Kaltschmidt, J. A., Yoshida, Y., Jessell, T. M. and Brownstone, R. M. (2005) Conditional rhythmicity of ventral spinal interneurons defined by expression of the Hb9 homeodomain protein. Journal of Neuroscience, 25(24), pp. 5710-5719. (doi: 10.1523/JNEUROSCI.0274-05.2005)
Hughes, D.I. et al. (2005) P boutons in lamina IX of the rodent spinal cord express high levels of glutamic acid decarboxylase-65 and originate from cells in deep medial dorsal horn. Proceedings of the National Academy of Sciences of the United States of America, 102(25), pp. 9038-9043. (doi: 10.1073/pnas.0503646102)
Alvarez, F., Jonas, P., Sapir, T., Hartley, R., Berrocal, M., Geiman, E., Todd, A. and Goulding, M. (2005) Postnatal phenotype and localization of spinal cord V1 derived interneurons. Journal of Comparative Neurology, 493, pp. 177-192. (doi: 10.1002/cne.20711)
Hinckley, C., Hartley, R., Wu, L., Todd, A. and Ziskind-Conhaim, L. (2005) Locomotor-like rhythms in a genetically distinct cluster of interneurons in the mammalian spinal cord. Journal of Neurophysiology, 93, pp. 1439-1449. (doi: 10.1152/jn.00647.2004)
Todd, A. , Spike, R., Young, S. and Puskar, Z. (2005) Fos induction in lamina I projection neurons in response to noxious thermal stimuli. Neuroscience, 131, pp. 209-217. (doi: 10.1016/j.neuroscience.2004.11.001)
Worsley, M., Todd, A. and King, A. (2005) Serotoninergic-mediated inhibition of substance P sensitive deep dorsal horn neurons: a combined electrophysiological and morphological study in vitro. Experimental Brain Research, 160, pp. 360-367. (doi: 10.1007/s00221-004-2018-7)
2004
Hughes, D.I. , Polgár, E., Shehab, S.A.S. and Todd, A.J. (2004) Peripheral axotomy induces depletion of the vesicular glutamate transporter VGLUT1 in central terminals of myelinated afferent fibres in the rat spinal cord. Brain Research, 1017(1-2), pp. 69-76. (doi: 10.1016/j.brainres.2004.05.054)
Halstead, S., OHanlon, G., Humphreys, P., Morrison, D., Morgan, B., Todd, A., Plomp, J. and Willison, H. (2004) Anti-disialoside antibodies kill perisynaptic Schwann cells and damage motor nerve terminals via membrane attack complex in a murine model of neuropathy. Brain, 127, pp. 2109-2123. (doi: 10.1093/brain/awh231)
Nagy, G., Al-Ayyan, M., Andrew, D., Fukaya, M., Watanabe, M. and Todd, A. (2004) Widespread expression of the AMPA receptor GluR2 subunit at glutamatergic synapses in the rat spinal cord and phosphorylation of GluR1 in response to noxious stimulation revealed with an antigen-unmasking method. Journal of Neuroscience, 24, pp. 5766-5777. (doi: 10.1523/JNEUROSCI.1237-04.2004)
Nagy, G., Watanabe, M., Fukaya, M. and Todd, A. (2004) Synaptic distribution of the NR1, NR2A and NR2B subunits of the N-methyl-D-aspartate receptor in the rat lumbar spinal cord revealed with an antigen-unmasking technique. European Journal of Neuroscience, 20, pp. 3301-3312. (doi: 10.1111/j.1460-9568.2004.03798.x)
Polgár, E., Gray, S., Riddell, J. and Todd, A. (2004) Lack of evidence for significant neuronal loss in laminae I-III of the spinal dorsal horn of the rat in the chronic constriction injury model. Pain, 111, pp. 144-150. (doi: 10.1016/j.pain.2004.06.011)
Shehab, S., Spike, R. and Todd, A. (2004) Do central terminals of intact myelinated primary afferents sprout into the superficial dorsal horn of rat spinal cord after injury to a neighboring peripheral nerve? Journal of Comparative Neurology, 474, pp. 427-437. (doi: 10.1002/cne.20147)
2003
Hughes, D.I. , Scott, D.T., Todd, A.J. and Riddell, J.S. (2003) Lack of evidence for sprouting of Aβ afferents into the superficial laminas of the spinal cord dorsal horn after nerve section. Journal of Neuroscience, 23(29), pp. 9491-9499.
Polgár, E., Hughes, D.I. , Riddell, J.S. , Maxwell, D.J. , Puskár, Z. and Todd, A.J. (2003) Selective loss of spinal GABAergic or glycinergic neurons is not necessary for development of thermal hyperalgesia in the chronic constriction injury model of neuropathic pain. Pain, 104(1), pp. 229-239. (doi: 10.1016/S0304-3959(03)00011-3)
Todd, A.J. , Hughes, D.I. , Polgár, E., Nagy, G.G., Mackie, M., Ottersen, O.P. and Maxwell, D.J. (2003) The expression of vesicular glutamate transporters VGLUT1 and VGLUT2 in neurochemically defined axonal populations in the rat spinal cord with emphasis on the dorsal horn. European Journal of Neuroscience, 17(1), pp. 13-27. (doi: 10.1046/j.1460-9568.2003.02406.x)
Hughes, D., Scott, D., Todd, A. and Riddell, J. (2003) Lack of evidence for sprouting of A beta afferents into the superficial laminas of the spinal cord dorsal horn after nerve section. Journal of Neuroscience, 23, pp. 9491-9499.
Mackie, M., Hughes, D.I. , Maxwell, D.J. , Tillakaratne, N.J.K. and Todd, A.J. (2003) Distribution and colocalisation of glutamate decarboxylase isoforms in the rat spinal cord. Neuroscience Letters, 119, pp. 461-472. (doi: 10.1016/S0306-4522(03)00174-X)
Shehab, S., Spike, R. and Todd, A. (2003) Evidence against cholera toxin B subunit as a reliable tracer for sprouting of primary afferents following peripheral nerve injury. Brain Research, 964, pp. 218-227. (doi: 10.1016/S0006-8993(02)04001-5)
Spike, R., Puskar, Z., Andrew, D. and Todd, A. (2003) A quantitative and morphological study of projection neurons in lamina I of the rat lumbar spinal cord. European Journal of Neuroscience, 18, pp. 2433-2448. (doi: 10.1046/j.1460-9568.2003.02981.x)
2002
Polgár, E., Puskar, Z., Watt, C., Matesz, C. and Todd, A. (2002) Selective innervation of lamina I projection neurones that possess the neurokinin 1 receptor by serotonin-containing axons in the rat spinal cord. Neuroscience, 109, pp. 799-809.
Spike, R.C., Puskar, Z., Sakamoto, H., Stewart, W. , Watt, C. and Todd, A.J. (2002) MOR-1-immunoreactive neurons in the dorsal horn of the rat spinal cord: evidence for nonsynaptic innervation by substance P-containing primary afferents and for selective activation by noxious thermal stimuli. European Journal of Neuroscience, 15(8), pp. 1306-1316. (doi: 10.1046/j.1460-9568.2002.01969.x)
Todd, A. (2002) Anatomy of primary afferents and projection neurones in the rat spinal dorsal horn with particular emphasis on substance P and the neurokinin 1 receptor. Experimental Physiology, 87, pp. 245-249.
Todd, A. , Puskar, Z., Spike, R., Hughes, C., Watt, C. and Forrest, L. (2002) Projection neurons in lamina I of rat spinal cord with the neurokinin 1 receptor are selectively innervated by substance p-containing afferents and respond to noxious stimulation. Journal of Neuroscience, 22, pp. 4103-4113.
2001
Puskar, Z., Polgár, E. and Todd, A. (2001) A population of large lamina I projection neurons with selective inhibitory input in rat spinal cord. Neuroscience, 102, pp. 167-176.
Articles
Quillet, R., Gutierrez-Mecinas, M. , Polgár, E., Dickie, A. C., Boyle, K. A., Watanabe, M. and Todd, A. J. (2023) Synaptic circuits involving gastrin-releasing peptide receptor-expressing neurons in the dorsal horn of the mouse spinal cord. Frontiers in Molecular Neuroscience, 16, 1294994. (doi: 10.3389/fnmol.2023.1294994) (PMID:38143564) (PMCID:PMC10742631)
Boyle, K. A. et al. (2023) Neuropeptide Y-expressing dorsal horn inhibitory interneurons gate spinal pain and itch signalling. eLife, 12, RP86633. (doi: 10.7554/eLife.86633) (PMID:37490401) (PMCID:PMC10392120)
Davis, O. C. et al. (2023) Calretinin-expressing islet cells are a source of pre- and post-synaptic inhibition of non-peptidergic nociceptor input to the mouse spinal cord. Scientific Reports, 13, 11561. (doi: 10.1038/s41598-023-38605-9) (PMID:37464016) (PMCID:PMC10354228)
Quillet, R., Dickie, A. C., Polgár, E., Gutierrez-Mecinas, M. , Bell, A. M. , Goffin, L., Watanabe, M. and Todd, A. J. (2023) Characterisation of NPFF-expressing neurons in the superficial dorsal horn of the mouse spinal cord. Scientific Reports, 13, 5891. (doi: 10.1038/s41598-023-32720-3) (PMID:37041197) (PMCID:PMC10090074)
Gutierrez-Mecinas, M. , Kókai, É., Polgár, E., Quillet, R., Titterton, H. F., Weir, G. A. , Watanabe, M. and Todd, A. J. (2023) Antibodies against the gastrin-releasing peptide precursor pro-gastrin-releasing peptide reveal its expression in the mouse spinal dorsal horn. Neuroscience, 510, pp. 60-71. (doi: 10.1016/j.neuroscience.2022.12.023) (PMID:36581131)
Ronzano, R. et al. (2023) Spinal premotor interneurons controlling antagonistic muscles are spatially intermingled. eLife, 11, e81976. (doi: 10.7554/elife.81976) (PMID:36512397) (PMCID:PMC9844990)
Polgár, E. et al. (2023) Grpr expression defines a population of superficial dorsal horn vertical cells that have a role in both itch and pain. Pain, 164(1), pp. 149-170. (doi: 10.1097/j.pain.0000000000002677) (PMID:35543635) (PMCID:PMC9756441)
Trendafilova, T. et al. (2022) Sodium-calcium exchanger-3 regulates pain "wind-up": from human psychophysics to spinal mechanisms. Neuron, 110(16), 2571-2587.e13. (doi: 10.1016/j.neuron.2022.05.017) (PMID:35705078) (PMCID:PMC7613464)
El Khoueiry, C., Alba-Delgado, C., Antri, M., Gutierrez-Mecinas, M. , Todd, A. J. , Artola, A. and Dallel, R. (2022) GABAA and glycine receptor-mediated inhibitory synaptic transmission onto adult rat lamina IIi PKCγ-interneurons: pharmacological but not anatomical specialization. Cells, 11(8), 1356. (doi: 10.3390/cells11081356) (PMID:35456035) (PMCID:PMC9033052)
Todd, A. J. (2022) An historical perspective: the second order neuron in the pain pathway. Frontiers in Pain Research, 3, 845211. (doi: 10.3389/fpain.2022.845211) (PMID:35295811) (PMCID:PMC8915774)
Kókai, É., Alsulaimain, W. A.A., Dickie, A. C., Bell, A. M. , Goffin, L., Watanabe, M., Gutierrez-Mecinas, M. and Todd, A. J. (2022) Characterisation of deep dorsal horn projection neurons in the spinal cord of the Phox2a::Cre mouse line. Molecular Pain, 18, p. 17448069221119614. (doi: 10.1177/17448069221119614) (PMID:36000342) (PMCID:PMC9445510)
Alsulaiman, W. A.A. et al. (2021) Characterisation of lamina I anterolateral system neurons that express Cre in a Phox2a-Cre mouse line. Scientific Reports, 11, 17912. (doi: 10.1038/s41598-021-97105-w) (PMID:34504158) (PMCID:PMC8429737)
Chisholm, K. I., Lo Re, L., Polgár, E., Gutierrez-Mecinas, M. , Todd, A. J. and McMahon, S. B. (2021) Encoding of cutaneous stimuli by lamina I projection neurons. Pain, 162(9), pp. 2405-2417. (doi: 10.1097/j.pain.0000000000002226) (PMID:33769365) (PMCID:PMC8374708)
Polgár, E., Bell, A. M. , Gutierrez-Mecinas, M. , Dickie, A. C., Akar, O., Costreie, M., Watanabe, M. and Todd, A. J. (2020) Substance P-expressing neurons in the superficial dorsal horn of the mouse spinal cord: insights into their functions and their roles in synaptic circuits. Neuroscience, 450, pp. 113-125. (doi: 10.1016/j.neuroscience.2020.06.038) (PMID:32634530)
Bell, A. M. , Gutierrez-Mecinas, M. , Stevenson, A., Casas-Benito, A., Wildner, H., West, S. J., Watanabe, M. and Todd, A. J. (2020) Expression of green fluorescent protein defines a specific population of lamina II excitatory interneurons in the GRP::eGFP mouse. Scientific Reports, 10, 13176. (doi: 10.1038/s41598-020-69711-7) (PMID:32764601) (PMCID:PMC7411045)
Hughes, D. I. and Todd, A. (2020) Central nervous system targets: inhibitory interneurons in the spinal cord. Neurotherapeutics, 17(3), pp. 874-885. (doi: 10.1007/s13311-020-00936-0) (PMID:33029722)
Tran, M., Braz, J. M., Hamel, K., Kuhn, J., Todd, A. J. and Basbaum, A. I. (2020) Ablation of spinal cord estrogen receptor α-expressing interneurons reduces chemically induced modalities of pain and itch. Journal of Comparative Neurology, 528(10), pp. 1629-1643. (doi: 10.1002/cne.24847) (PMID:31872868) (PMCID:PMC7317200)
Peirs, C., Dallel, R. and Todd, A. J. (2020) Recent advances in our understanding of the organization of dorsal horn neuron populations and their contribution to cutaneous mechanical allodynia. Journal of Neural Transmission, 127, pp. 505-525. (doi: 10.1007/s00702-020-02159-1) (PMID:32239353) (PMCID:PMC7148279)
Gutierrez-Mecinas, M. , Bell, A. M. , Shepherd, F., Polgár, E., Watanabe, M., Furuta, T. and Todd, A. J. (2019) Expression of cholecystokinin by neurons in mouse spinal dorsal horn. Journal of Comparative Neurology, 527(11), pp. 1857-1871. (doi: 10.1002/cne.24657) (PMID:30734936) (PMCID:PMC6563475)
Dickie, A. C. et al. (2019) Morphological and functional properties distinguish the substance P and gastrin-releasing peptide subsets of excitatory interneuron in the spinal cord dorsal horn. Pain, 160(2), pp. 442-462. (doi: 10.1097/j.pain.0000000000001406) (PMID:30247267) (PMCID:PMC6330098)
Gutierrez-Mecinas, M. , Davis, O., Polgár, E., Shahzad, M., Navarro-Batista, K., Furuta, T., Watanabe, M., Hughes, D. I. and Todd, A. J. (2019) Expression of calretinin among different neurochemical classes of interneuron in the superficial dorsal horn of the mouse spinal cord. Neuroscience, 398, pp. 171-181. (doi: 10.1016/j.neuroscience.2018.12.009) (PMID:30553791) (PMCID:PMC6347472)
Gutierrez-Mecinas, M. , Bell, A. , Polgár, E., Watanabe, M. and Todd, A. J. (2019) Expression of neuropeptide FF defines a population of excitatory interneurons in the superficial dorsal horn of the mouse spinal cord that respond to noxious and pruritic stimuli. Neuroscience, 416, pp. 281-293. (doi: 10.1016/j.neuroscience.2019.08.013) (PMID:31421202) (PMCID:PMC6839401)
Gutierrez-Mecinas, M. , Polgár, E., Bell, A. M. , Hérau, M. and Todd, A. J. (2018) Substance P-expressing excitatory interneurons in the mouse superficial dorsal horn provide a propriospinal input to the lateral spinal nucleus. Brain Structure and Function, 223(5), pp. 2377-2392. (doi: 10.1007/s00429-018-1629-x) (PMID:29497838) (PMCID:PMC5968060)
Huang, J. et al. (2018) Author correction: circuit dissection of the role of somatostatin in itch and pain. Nature Neuroscience, 21(6), p. 894. (doi: 10.1038/s41593-018-0149-6) (PMID:29674654)
Strnadel, J. et al. (2018) Survival of syngeneic and allogeneic iPSC–derived neural precursors after spinal grafting in minipigs. Science Translational Medicine, 10(440), eaam6651. (doi: 10.1126/scitranslmed.aam6651) (PMID:29743351)
Huang, J. et al. (2018) Circuit dissection of the role of somatostatin in itch and pain. Nature Neuroscience, 21(5), pp. 707-716. (doi: 10.1038/s41593-018-0119-z) (PMID:29556030) (PMCID:PMC5923877)
Dawes, J. M. et al. (2018) Immune or genetic-mediated disruption of CASPR2 causes pain hypersensitivity due to enhanced primary afferent excitability. Neuron, 97(4), 806-822.e10. (doi: 10.1016/j.neuron.2018.01.033) (PMID:29429934)
Boyle, K. A., Gutierrez-Mecinas, M. , Polgár, E., Mooney, N., O'Connor, E., Furuta, T., Watanabe, M. and Todd, A. J. (2017) A quantitative study of neurochemically-defined populations of inhibitory interneurons in the superficial dorsal horn of the mouse spinal cord. Neuroscience, 363, pp. 120-133. (doi: 10.1016/j.neuroscience.2017.08.044) (PMID:28860091) (PMCID:PMC5648048)
Gutierrez-Mecinas, M. , Bell, A. M. , Marin, A., Taylor, R., Boyle, K. A., Furuta, T., Watanabe, M., Polgár, E. and Todd, A. J. (2017) Preprotachykinin A (PPTA) is expressed by a distinct population of excitatory neurons in the mouse superficial spinal dorsal horn including cells that respond to noxious and pruritic stimuli. Pain, 158(3), pp. 440-456. (doi: 10.1097/j.pain.0000000000000778) (PMID:27902570) (PMCID:PMC5302415)
Todd, A. J. (2017) Identifying functional populations among the interneurons in laminae I-III of the spinal dorsal horn. Molecular Pain, 13, pp. 1-19. (doi: 10.1177/1744806917693003) (PMID:28326935) (PMCID:PMC5315367)
Bell, A. M. , Gutierrez-Mecinas, M. , Polgár, E. and Todd, A. J. (2016) Spinal neurons that contain gastrin-releasing peptide seldom express Fos or phosphorylate extracellular signal-regulated kinases in response to intradermal chloroquine. Molecular Pain, 12, pp. 1-9. (doi: 10.1177/1744806916649602) (PMID:27270268) (PMCID:PMC4937990)
Gutierrez-Mecinas, M. , Furuta, T., Watanabe, M. and Todd, A. J. (2016) A quantitative study of neurochemically-defined excitatory interneuron populations in laminae I-III of the mouse spinal cord. Molecular Pain, 12, pp. 1-18. (doi: 10.1177/1744806916629065) (PMID:27030714) (PMCID:PMC4946630)
Gutierrez-Mecinas, M. , Kuehn, E. D., Abraira, V. E., Polgár, E., Watanabe, M. and Todd, A. J. (2016) Immunostaining for Homer reveals the majority of excitatory synapses in laminae I-III of the mouse spinal dorsal horn. Neuroscience, 329, pp. 171-181. (doi: 10.1016/j.neuroscience.2016.05.009) (PMID:27185486) (PMCID:PMC4915440)
Chiang, M.C., Hachisuka, J. , Todd, A.J. and Ross, S.E. (2016) Erratum. Pain, 157(4), p. 992. (doi: 10.1097/j.pain.0000000000000552) (PMID:26982518)
Chiang, M. C., Hachisuka, J. , Todd, A. J. and Ross, S. E. (2016) Insight into B5-I spinal interneurons and their role in the inhibition of itch and pain. Pain, 157(3), pp. 544-545. (doi: 10.1097/j.pain.0000000000000474) (PMID:26716996)
Iwagaki, N., Ganley, R. P., Dickie, A., Polgár, E., Hughes, D. I. , Del Rio, P., Revina, Y., Watanabe, M., Todd, A. J. and Riddell, J. S. (2016) A combined electrophysiological and morphological study of NPY-expressing inhibitory interneurons in the spinal dorsal horn of the mouse. Pain, 157(3), pp. 598-612. (doi: 10.1097/j.pain.0000000000000407) (PMID:26882346) (PMCID:PMC4751741)
Ganley, R. P. et al. (2015) Inhibitory interneurons that express GFP in the PrP-GFP mouse spinal cord are morphologically heterogeneous, innervated by several classes of primary afferent and include lamina I projection neurons among their postsynaptic targets. Journal of Neuroscience, 35(19), pp. 7626-7642. (doi: 10.1523/JNEUROSCI.0406-15.2015) (PMID:25972186) (PMCID:PMC4429159)
Cameron, D., Gutierrez-Mecinas, M. , Gomez-Lima, M., Watanabe, M., Polgár, E. and Todd, A. J. (2015) The organisation of spinoparabrachial neurons in the mouse. Pain, 156(10), pp. 2061-2071. (doi: 10.1097/j.pain.0000000000000270) (PMID:26101837) (PMCID:PMC4770364)
Baseer, N., Al-Baloushi, A. S., Watanabe, M., Shehab, S. A.S. and Todd, A. J. (2014) Selective innervation of NK1 receptor–lacking lamina I spinoparabrachial neurons by presumed nonpeptidergic Aδ nociceptors in the rat. Pain, 155(11), pp. 2291-2300. (doi: 10.1016/j.pain.2014.08.023) (PMID:25168670) (PMCID:PMC4247378)
Bhumbra, G. S., Bannatyne, B. A., Watanabe, M., Todd, A. J. , Maxwell, D. J. and Beato, M. (2014) The recurrent case for the Renshaw cell. Journal of Neuroscience, 34(38), pp. 12919-12932. (doi: 10.1523/JNEUROSCI.0199-14.2014) (PMID:25232126) (PMCID:PMC4166169)
Kardon, A. P. et al. (2014) Dynorphin acts as a neuromodulator to inhibit itch in the dorsal horn of the spinal cord. Neuron, 82(3), pp. 573-586. (doi: 10.1016/j.neuron.2014.02.046) (PMID:24726382) (PMCID:PMC4022838)
Gutierrez-Mecinas, M. , Watanabe, M. and Todd, A. J. (2014) Expression of gastrin-releasing peptide by excitatory interneurons in the mouse superficial dorsal horn. Molecular Pain, 10, 79. (doi: 10.1186/1744-8069-10-79) (PMID:25496164) (PMCID:PMC4320531)
Yasaka, T., Tiong, S. Y.X., Polgár, E., Watanabe, M., Kumamoto, E., Riddell, J. S. and Todd, A. J. (2014) A putative relay circuit providing low-threshold mechanoreceptive input to lamina I projection neurons via vertical cells in lamina II of the rat dorsal horn. Molecular Pain, 10(1), p. 3. (doi: 10.1186/1744-8069-10-3)
Polgár, E., Sardella, T.C.P., Tiong, S.Y.X., Locke, S., Watanabe, M. and Todd, A.J. (2013) Functional differences between neurochemically defined populations of inhibitory interneurons in the rat spinal dorsal horn. Pain, 154(12), pp. 2606-2615. (doi: 10.1016/j.pain.2013.05.001)
Szucs, P., Luz, L.L., Pinho, R., Aguiar, P., Antal, Z., Tiong, S.Y.X., Todd, A.J. and Safronov, B.V. (2013) Axon diversity of lamina I local-circuit neurons in the lumbar spinal cord. Journal of Comparative Neurology, 521(12), pp. 2719-2741. (doi: 10.1002/cne.23311)
Iwagaki, N., Garzillo, F., Polgár, E., Riddell, J.S. and Todd, A.J. (2013) Neurochemical characterisation of lamina II inhibitory interneurons that express GFP in the PrP-GFP mouse. Molecular Pain, 9(56), (doi: 10.1186/1744-8069-9-56)
Polgár, E., Durrieux, C., Hughes, D.I. and Todd, A.J. (2013) A quantitative study of inhibitory interneurons in laminae I-III of the mouse spinal dorsal horn. PLoS ONE, 8(10), e78309. (doi: 10.1371/journal.pone.0078309) (PMID:24205193) (PMCID:PMC3808353)
Baseer, N., Watanabe, M., Furuta, T., Kaneko, T. and Todd, A.J. (2012) Projection neurons in lamina III of the rat spinal cord are selectively innervated by local dynorphin-containing excitatory neurons. Journal of Neuroscience, 32(34), pp. 11854-11863. (doi: 10.1523/JNEUROSCI.2707-12.2012)
Tappe-Theodor, A. et al. (2012) Gαq/11 signaling tonically modulates nociceptor function and contributes to activity-dependent sensitization. Pain, 153(1), pp. 184-196. (doi: 10.1016/j.pain.2011.10.014)
Baseer, N., Polgar, E., Watanabe, M., Furuta, T., Kaneko, T. and Todd, A. (2012) Projection neurons in Lamina III of the rat spinal cord are selectively innervated by local dynorphin-containing excitatory neurons. Journal of Neuroscience, 32(34), pp. 11854-11863. (doi: 10.1523/JNEUROSCI.2707-12.2012)
Luo, C. et al. (2012) Presynaptically localized cyclic GMP-dependent protein kinase 1 is a key determinant of spinal synaptic potentiation and pain hypersensitivity. PLoS Biology, 10(3), e1001283. (doi: 10.1371/journal.pbio.1001283)
Todd, A.J. (2012) How to recognise collateral damage in partial nerve injury models of neuropathic pain. Pain, 153(1), pp. 11-12. (doi: 10.1016/j.pain.2011.10.031)
Tiong, S. Y.X., Polgár, E., van Kralingen, J. C., Watanabe, M. and Todd, A. J. (2011) Galanin-immunoreactivity identifies a distinct population of inhibitory interneurons in laminae I-III of the rat spinal cord. Molecular Pain, 7, 36. (doi: 10.1186/1744-8069-7-36) (PMID:21569622) (PMCID:PMC3118366)
Polgár, E., Sardella, T.C.P., Watanabe, M. and Todd, A.J. (2011) Quantitative study of NPY-expressing GABAergic neurons and axons in rat spinal dorsal horn. Journal of Comparative Neurology, 519(6), pp. 1007-1023. (doi: 10.1002/cne.22570)
Saywell, S.A., Ford, T.W., Meehan, C.F., Todd, A.J. and Kirkwood, P.A. (2011) Electrophysiological and morphological characterization of propriospinal interneurons in the thoracic spinal cord. Journal of Neurophysiology, 105(2), pp. 806-826. (doi: 10.1152/jn.00738.2010)
Gangadharan, V. et al. (2011) Peripheral calcium-permeable AMPA receptors regulate chronic inflammatory pain in mice. Journal of Clinical Investigation, 121(4), pp. 1608-1623. (doi: 10.1172/JCI44911)
Sardella, T.C.P., Polgár, E., Garzillo, F., Furuta, T., Kaneko, T., Watanabe, M. and Todd, A.J. (2011) Dynorphin is expressed primarily by GABAergic neurons that contain galanin in the rat dorsal horn. Molecular Pain, 7(1), p. 76. (doi: 10.1186/1744-8069-7-76)
Sardella, T.C.P., Polgár, E., Watanabe, M. and Todd, A.J. (2011) A quantitative study of neuronal nitric oxide synthase expression in laminae I–III of the rat spinal dorsal horn. Neuroscience, 192, pp. 708-720. (doi: 10.1016/j.neuroscience.2011.07.011)
Todd, A.J. (2010) Neuronal circuitry for pain processing in the dorsal horn. Nature Reviews Neuroscience, 11(12), pp. 823-836. (doi: 10.1038/nrn2947)
Yasaka, T., Tiong, S.Y.X., Hughes, D.I. , Riddell, J.S. and Todd, A.J. (2010) Populations of inhibitory and excitatory interneurons in lamina II of the adult rat spinal dorsal horn revealed by a combined electrophysiological and anatomical approach. Pain, 151(2), pp. 475-488. (doi: 10.1016/j.pain.2010.08.008)
Polgár, E., Wright, L. L. and Todd, A. J. (2010) A quantitative study of brainstem projections from lamina I neurons in the cervical and lumbar enlargement of the rat. Brain Research, 1308, pp. 58-67. (doi: 10.1016/j.brainres.2009.10.041)
Polgár, E., Al Ghamdi, K.S. and Todd, A.J. (2010) Two populations of neurokinin 1 receptor-expressing projection neurons in lamina I of the rat spinal cord that differ in AMPA receptor subunit composition and density of excitatory synaptic input. Neuroscience, 167(4), pp. 1192-1204. (doi: 10.1016/j.neuroscience.2010.03.028)
Al Ghamdi, K.S., Polgár, E. and Todd, A. (2009) Soma size distinguishes projection neurons from neurokinin 1 receptor-expressing interneurons in lamina I of the rat lumbar spinal dorsal horn. Neuroscience, 164(4), pp. 1794-1804. (doi: 10.1016/j.neuroscience.2009.09.071)
Yasaka, T., Hughes, D.I. , Polgár, E., Nagy, G.G., Watanabe, M., Riddell, J.S. and Todd, A.J. (2009) Evidence against AMPA receptor-lacking glutamatergic synapses in the superficial dorsal horn of the rat spinal cord. Journal of Neuroscience, 29(42), pp. 13401-13409. (doi: 10.1523/JNEUROSCI.2628-09.2009)
Todd, A. J. , Polgár, E., Watt, C., Bailey, M. E.S. and Watanabe, M. (2009) Neurokinin 1 receptor-expressing projection neurons in laminae III and IV of the rat spinal cord have synaptic AMPA receptors that contain GluR2, GluR3 and GluR4 subunits. European Journal of Neuroscience, 29(4), pp. 718-726. (doi: 10.1111/j.1460-9568.2009.06633.x) (PMID:19200070) (PMCID:PMC2695158)
Al-Khater, K.M. and Todd, A.J. (2009) Collateral projections of neurons in laminae I, III, and IV of rat spinal cord to thalamus, periaqueductal gray matter, and lateral parabrachial area. Journal of Comparative Neurology, 515(6), pp. 629-646. (doi: 10.1002/cne.22081)
Polgar, E., Al-Khater, K.M., Shehab, S., Watanabe, M. and Todd, A.J. (2008) Large projection neurons in lamina I of the rat spinal cord that lack the neurokinin 1 receptor are densely innervated by VGLUT2-containing axons and possess GluR4-containing AMPA receptors. Journal of Neuroscience, 28(49), pp. 13150-13160. (doi: 10.1523/JNEUROSCI.4053-08.2008)
Polgár, E. and Todd, A. (2008) Tactile allodynia can occur in the spared nerve injury model in the rat without selective loss of GABA or GABAA receptors from synapses in laminae I–II of the ipsilateral spinal dorsal horn. Neuroscience, 156(1), pp. 193-202. (doi: 10.1016/j.neuroscience.2008.07.009)
Polgár, E., Watanabe, M., Hartmann, B., Grant, S. and Todd, A.J. (2008) Expression of AMPA receptor subunits at synapses in laminae I-III of the rodent spinal dorsal horn. Molecular Pain, 4(5), pp. 1-14. (doi: 10.1186/1744-8069-4-5)
Al-Khater, K.M., Kerr, R. and Todd, A.J. (2008) A quantitative study of spinothalamic neurons in laminae I, III, and IV in lumbar and cervical segments of the rat spinal cord. Journal of Comparative Neurology, 511(1), pp. 1-18. (doi: 10.1002/cne.21811)
Hughes, D.I. , Scott, D.T., Riddell, J.S. and Todd, A.J. (2007) Up-regulation of substance P in low-threshold myelinated afferents is not required for tactile allodynia in the chronic constriction injury and spinal nerve ligation models. Journal of Neuroscience, 27(8), pp. 2035-2044. (doi: 10.1523/JNEUROSCI.5401-06.2007)
Marvizón, J.C.G., Pérez, O.A., Song, B., Chen, W., Bunnett, N.W., Grady, E.F. and Todd, A.J. (2007) Calcitonin receptor-like receptor and receptor activity modifying protein 1 in the rat dorsal horn: localization in glutamatergic presynaptic terminals containing opioids and adrenergic α2C receptors. Journal of Neuroscience, 148(1), pp. 250-265. (doi: 10.1016/j.neuroscience.2007.05.036)
Miles, G.B., Hartley, R., Todd, A.J. and Brownstone, R.M. (2007) Spinal cholinergic interneurons regulate the excitability of motoneurons during locomotion. Proceedings of the National Academy of Sciences of the United States of America, 104(7), pp. 2448-2453.
Polgár, E., Campbell, A.D., MacIntyre, L., Watanabe, M. and Todd, A.J. (2007) Phosphorylation of ERK in neurokinin 1 receptor-expressing neurons in laminae III and IV of the rat spinal dorsal horn following noxious stimulation. Molecular Pain, 3(4),
Hughes, D.I. , Scott, D.T., Riddell, J.S. and Todd, A.J. (2007) Upregulation of substance P in low-threshold myelinated afferents is not required for tactile allodynia in the chronic constriction injury and spinal nerve ligation models. Journal of Neuroscience, 27(8), pp. 2035-2044. (doi: 10.1523/JNEUROSCI.5401-06.2007)
Marvizon, J., Perez, O., Song, B., Chen, W., Bunnett, N., Grady, E. and Todd, A. (2007) Calcitonin receptor-like and receptor activity modifying protein 1 in the rat dorsal horn localization in glutamatergic presynaptic terminal containing opioids and adrenergic alpha(2c) receptor. Neuroscience, 148, pp. 250-265. (doi: 10.1016/j.neuroscience.2007.05.036)
Polgar, E., Thomson, S., Maxwell, D., Al-Khater, K. and Todd, A. (2007) A population of large neurons in laminae III and IV of the rat spinal cord that have long dorsal dendrites and lack the neurokinin 1 receptor. European Journal of Neuroscience, 26, pp. 1587-1598. (doi: 10.1111/j.1460-9568.2007.05793.x)
Polgar, E., Furuta, T., Kaneko, T. and Todd, A. (2006) Characterization of neurons that express preprotachykinin B in the dorsal horn of the rat spinal cord. Neuroscience, 139, pp. 687-697. (doi: 10.1016/j.neuroscience.2005.12.021)
Todd, A. (2006) Erratum to 'Characterization of neurons that express preprotachykinin B in the dorsal horn of the rat spinal cord': [Neuroscience 139 (2006) 687-697]. Neuroscience, 141(4), pp. 2195-2197. (doi: 10.1016/j.neuroscience.2005.12.0)
Shehab, S., McGonigle, D., Hughes, D.I. , Todd, A.J. and Redgrave, P. (2005) Anatomical evidence for an anticonvulsant relay in the rat ventromedial medulla. European Journal of Neuroscience, 22(6), pp. 1431-1444. (doi: 10.1111/j.1460-9568.2005.04326.x)
Polgar, E., Hughes, D.I. , Arham, A.Z. and Todd, A.J. (2005) Loss of neurons from laminas I-III of the spinal dorsal horn is not required for development of tactile allodynia in the spared nerve injury model of neuropathic pain. Journal of Neuroscience, 25(28), pp. 6658-6666. (doi: 10.1523/JNEUROSCI.1490-05.2005)
Wilson, J. W., Hartley, R., Maxwell, D. J. , Todd, A. J. , Lieberam, I., Kaltschmidt, J. A., Yoshida, Y., Jessell, T. M. and Brownstone, R. M. (2005) Conditional rhythmicity of ventral spinal interneurons defined by expression of the Hb9 homeodomain protein. Journal of Neuroscience, 25(24), pp. 5710-5719. (doi: 10.1523/JNEUROSCI.0274-05.2005)
Hughes, D.I. et al. (2005) P boutons in lamina IX of the rodent spinal cord express high levels of glutamic acid decarboxylase-65 and originate from cells in deep medial dorsal horn. Proceedings of the National Academy of Sciences of the United States of America, 102(25), pp. 9038-9043. (doi: 10.1073/pnas.0503646102)
Alvarez, F., Jonas, P., Sapir, T., Hartley, R., Berrocal, M., Geiman, E., Todd, A. and Goulding, M. (2005) Postnatal phenotype and localization of spinal cord V1 derived interneurons. Journal of Comparative Neurology, 493, pp. 177-192. (doi: 10.1002/cne.20711)
Hinckley, C., Hartley, R., Wu, L., Todd, A. and Ziskind-Conhaim, L. (2005) Locomotor-like rhythms in a genetically distinct cluster of interneurons in the mammalian spinal cord. Journal of Neurophysiology, 93, pp. 1439-1449. (doi: 10.1152/jn.00647.2004)
Todd, A. , Spike, R., Young, S. and Puskar, Z. (2005) Fos induction in lamina I projection neurons in response to noxious thermal stimuli. Neuroscience, 131, pp. 209-217. (doi: 10.1016/j.neuroscience.2004.11.001)
Worsley, M., Todd, A. and King, A. (2005) Serotoninergic-mediated inhibition of substance P sensitive deep dorsal horn neurons: a combined electrophysiological and morphological study in vitro. Experimental Brain Research, 160, pp. 360-367. (doi: 10.1007/s00221-004-2018-7)
Hughes, D.I. , Polgár, E., Shehab, S.A.S. and Todd, A.J. (2004) Peripheral axotomy induces depletion of the vesicular glutamate transporter VGLUT1 in central terminals of myelinated afferent fibres in the rat spinal cord. Brain Research, 1017(1-2), pp. 69-76. (doi: 10.1016/j.brainres.2004.05.054)
Halstead, S., OHanlon, G., Humphreys, P., Morrison, D., Morgan, B., Todd, A., Plomp, J. and Willison, H. (2004) Anti-disialoside antibodies kill perisynaptic Schwann cells and damage motor nerve terminals via membrane attack complex in a murine model of neuropathy. Brain, 127, pp. 2109-2123. (doi: 10.1093/brain/awh231)
Nagy, G., Al-Ayyan, M., Andrew, D., Fukaya, M., Watanabe, M. and Todd, A. (2004) Widespread expression of the AMPA receptor GluR2 subunit at glutamatergic synapses in the rat spinal cord and phosphorylation of GluR1 in response to noxious stimulation revealed with an antigen-unmasking method. Journal of Neuroscience, 24, pp. 5766-5777. (doi: 10.1523/JNEUROSCI.1237-04.2004)
Nagy, G., Watanabe, M., Fukaya, M. and Todd, A. (2004) Synaptic distribution of the NR1, NR2A and NR2B subunits of the N-methyl-D-aspartate receptor in the rat lumbar spinal cord revealed with an antigen-unmasking technique. European Journal of Neuroscience, 20, pp. 3301-3312. (doi: 10.1111/j.1460-9568.2004.03798.x)
Polgár, E., Gray, S., Riddell, J. and Todd, A. (2004) Lack of evidence for significant neuronal loss in laminae I-III of the spinal dorsal horn of the rat in the chronic constriction injury model. Pain, 111, pp. 144-150. (doi: 10.1016/j.pain.2004.06.011)
Shehab, S., Spike, R. and Todd, A. (2004) Do central terminals of intact myelinated primary afferents sprout into the superficial dorsal horn of rat spinal cord after injury to a neighboring peripheral nerve? Journal of Comparative Neurology, 474, pp. 427-437. (doi: 10.1002/cne.20147)
Hughes, D.I. , Scott, D.T., Todd, A.J. and Riddell, J.S. (2003) Lack of evidence for sprouting of Aβ afferents into the superficial laminas of the spinal cord dorsal horn after nerve section. Journal of Neuroscience, 23(29), pp. 9491-9499.
Polgár, E., Hughes, D.I. , Riddell, J.S. , Maxwell, D.J. , Puskár, Z. and Todd, A.J. (2003) Selective loss of spinal GABAergic or glycinergic neurons is not necessary for development of thermal hyperalgesia in the chronic constriction injury model of neuropathic pain. Pain, 104(1), pp. 229-239. (doi: 10.1016/S0304-3959(03)00011-3)
Todd, A.J. , Hughes, D.I. , Polgár, E., Nagy, G.G., Mackie, M., Ottersen, O.P. and Maxwell, D.J. (2003) The expression of vesicular glutamate transporters VGLUT1 and VGLUT2 in neurochemically defined axonal populations in the rat spinal cord with emphasis on the dorsal horn. European Journal of Neuroscience, 17(1), pp. 13-27. (doi: 10.1046/j.1460-9568.2003.02406.x)
Hughes, D., Scott, D., Todd, A. and Riddell, J. (2003) Lack of evidence for sprouting of A beta afferents into the superficial laminas of the spinal cord dorsal horn after nerve section. Journal of Neuroscience, 23, pp. 9491-9499.
Mackie, M., Hughes, D.I. , Maxwell, D.J. , Tillakaratne, N.J.K. and Todd, A.J. (2003) Distribution and colocalisation of glutamate decarboxylase isoforms in the rat spinal cord. Neuroscience Letters, 119, pp. 461-472. (doi: 10.1016/S0306-4522(03)00174-X)
Shehab, S., Spike, R. and Todd, A. (2003) Evidence against cholera toxin B subunit as a reliable tracer for sprouting of primary afferents following peripheral nerve injury. Brain Research, 964, pp. 218-227. (doi: 10.1016/S0006-8993(02)04001-5)
Spike, R., Puskar, Z., Andrew, D. and Todd, A. (2003) A quantitative and morphological study of projection neurons in lamina I of the rat lumbar spinal cord. European Journal of Neuroscience, 18, pp. 2433-2448. (doi: 10.1046/j.1460-9568.2003.02981.x)
Polgár, E., Puskar, Z., Watt, C., Matesz, C. and Todd, A. (2002) Selective innervation of lamina I projection neurones that possess the neurokinin 1 receptor by serotonin-containing axons in the rat spinal cord. Neuroscience, 109, pp. 799-809.
Spike, R.C., Puskar, Z., Sakamoto, H., Stewart, W. , Watt, C. and Todd, A.J. (2002) MOR-1-immunoreactive neurons in the dorsal horn of the rat spinal cord: evidence for nonsynaptic innervation by substance P-containing primary afferents and for selective activation by noxious thermal stimuli. European Journal of Neuroscience, 15(8), pp. 1306-1316. (doi: 10.1046/j.1460-9568.2002.01969.x)
Todd, A. (2002) Anatomy of primary afferents and projection neurones in the rat spinal dorsal horn with particular emphasis on substance P and the neurokinin 1 receptor. Experimental Physiology, 87, pp. 245-249.
Todd, A. , Puskar, Z., Spike, R., Hughes, C., Watt, C. and Forrest, L. (2002) Projection neurons in lamina I of rat spinal cord with the neurokinin 1 receptor are selectively innervated by substance p-containing afferents and respond to noxious stimulation. Journal of Neuroscience, 22, pp. 4103-4113.
Puskar, Z., Polgár, E. and Todd, A. (2001) A population of large lamina I projection neurons with selective inhibitory input in rat spinal cord. Neuroscience, 102, pp. 167-176.
Book Sections
Hughes, D. I. and Todd, A. J. (2022) Visualizing synaptic connectivity using confocal and electron microscopy: neuroanatomical approaches to define spinal circuits. In: Contemporary Approaches to the Study of Pain. Series: Neuromethods, 178. Humana: New York, pp. 181-201. ISBN 9781071620380 (doi: 10.1007/978-1-0716-2039-7_11)
Bell, A. M. , Polgár, E., Gutierrez-Mecinas, M. , Boyle, K. A. and Todd, A. J. (2020) Functional populations among interneurons in the dorsal horn. In: Fritzsch, B. (ed.) The Senses: A Comprehensive Reference. Second Edition. Academic Press: Cambridge, MA, pp. 207-219. ISBN 9780128054093 (doi: 10.1016/B978-0-12-809324-5.24171-0)
Todd, A. J. (2016) Anatomy of pain pathways. In: Battaglia, A. A. (ed.) An Introduction to Pain and its Relation to Nervous System Disorders. Wiley Blackwell, pp. 11-33. ISBN 9781118455913 (doi: 10.1002/9781118455968.ch1)
Todd, A. J. (2015) Plasticity of inhibition in the spinal cord. In: Schaible, H.-G. (ed.) Pain Control. Series: Handbook of experimental pharmacology. Springer, pp. 171-190. ISBN 9783662464496 (doi: 10.1007/978-3-662-46450-2_9)
Ross, S. E., Hachisuka, J. and Todd, A. J. (2014) Spinal microcircuits and the regulation of itch. In: Carstens, E. and Akiyama, T. (eds.) Itch: Mechanisms and Treatment. CRC Press Taylor & Francis Group. ISBN 9781466505438
Grants
Grants and Awards listed are those received whilst working with the University of Glasgow.
- Spinal modulation of non-peptidergic C-nociceptor input by inhibitory calretinin neurons
Biotechnology and Biological Sciences Research Council
2023 - 2025
- The roles of functionally defined populations of lamina I projection neurons
Medical Research Council
2022 - 2024
- Spinal circuits underlying pathological pain
Wellcome Trust
2020 - 2025
- Neuronal circuits for itch in the spinal dorsal horn
Medical Research Council
2018 - 2021
- Determining the role of calretinin-RorB spinal interneurons in modulating mechanical pain
Biotechnology and Biological Sciences Research Council
2017 - 2020
- The role of NPY-containing inhibitory interneurons in spinal pain pathways
Biotechnology and Biological Sciences Research Council
2016 - 2019
- Defining pain circuitry in health and disease
Wellcome Trust
2014 - 2019
- Pre-motor neuronal networks , from connectivity to function
Biotechnology and Biological Sciences Research Council
2013 - 2017
- Spinal inhibitory interneurons that suppress itch
Medical Research Council
2013 - 2016
- Populations of inhibitory interneurons in the superficial dorsal horn of the spinal cord.
Biotechnology and Biological Sciences Research Council
2012 - 2015
- Wellcome VIP
Wellcome Trust
2011 - 2011
- Confocal microscopic studies in Neuroscience
Wellcome Trust
2010 - 2013
- A quantitative description of glycinergic circuits in the ventral horn of the lumbar spinal cord.
Wellcome Trust
2009 - 2012
- The role of spinal GluR4 containing AMPA receptors in pain mechanisms
Tenovus Scotland
2007 - 2007
- AMPA receptors in the spinal dorsal horn - synaptic distribution and plasticity in pain states
Wellcome Trust
2006 - 2010
- P boutons in the rodent spinal cord
The Carnegie Trust for the Universities of Scotland
2005 - 2006
- Confocal microscopic studies in the spinal cord
Wellcome Trust
2002 - 2005
- An Investigation of Spinal Cord Mechanisms Underlying Neuropathic Pain in Nerve Injury Models in the Rat
Wellcome Trust
2000 - 2006
Supervision
Supervised Postgraduate Students
- Wafa Abdulsalam Alsulaiman (Postgraduate)
- Jane Brandon (Postgraduate)
- Paschalina Chrysostomidou (Postgraduate)
- Mai Hajer (Postgraduate)
- Wenhui Ma (Postgraduate)
- Aimi Nadia Razlan (Postgraduate)
Research Assistants
- Maria Gutierrez-Mecinas (Research Associate)
- Eva Kokai (Research Assistant)
- Raphaelle Quillet (Research Associate)
- Gregory Weir (Senior Research Fellow)
Visiting Collaborator
- Jane Brandon (Visiting Researcher)
Research datasets
2023
Todd, A. , Quillet, R., Gutierrez-Mecinas, M. , Boyle, K., Polgar, E., Dickie, A. and Riddell, J. (2023) Synaptic circuits involving gastrin-releasing peptide receptor-expressing neurons in the dorsal horn of the mouse spinal cord. [Data Collection]
Todd, A. , Boyle, K., Polgar, E., Gutierrez-Mecinas, M. , Dickie, A., Cooper, A., Bell, A., Jumolea, M. E., Casas-Benito, A., Watanabe, M., Hughes, D., Weir, G. and Riddell, J. (2023) Neuropeptide Y-expressing dorsal horn inhibitory interneurons gate spinal pain and itch signalling. [Data Collection]
Todd, A. , Polgar, E., Dickie, A., Gutierrez-Mecinas, M. , Bell, A. , Goffin, L., Quillet, R. and Watanabe, M. (2023) Characterisation of NPFF-expressing neurons in the superficial dorsal horn of the mouse spinal cord. [Data Collection]
2022
Todd, A. , Polgar, E., Dickie, A., Gutierrez-Mecinas, M. , Bell, A. , Boyle, K. and Quillet, R. (2022) Grpr expression defines a population of superficial dorsal horn vertical cells that have a role in both itch and pain. [Data Collection]
Additional information
Editorial Board
- 2009 - present: Frontiers in Neuroanatomy
- 2004 - present: Molecular Pain
- 2000 - present: Neuroscience
Grant Advisory Board
- 2009 - present: Wellcome Trust - Chairman of the External Advisory Board of the London Pain Consortium
Invited International Presentations
- 2011: Hamburg, Germany - Refresher course, EFIC meeting, invited speaker
- 2011: Heidleberg, Germany - EFIC Satellite symposium, speaker
- 2011: Porto, Portugal - Departmental Seminar
- 2009: Zurich, Switzerland - Invited seminar
- 2009: Nagoya, Japan - Plenary lecturer, "Nagoya Pain 2009", (joint meeting of Japanese Association for the Study of Pain and Japanese Society of Pain Clinicians)
- 2009: New York, USA - Plenary Lecture - World Institute of Pain (WIP) 5th World Congress
- 2009: El Escorial, Spain - Invited speaker, London Pain Consortium summer school
- 2009: Bordeaux, France - Symposium speaker, 9th Congress French Neuroscience Society
- 2008: Glasgow, UK - Plenary Lecture - International Association for the Study of Pain (IASP) 12th World Congress
- 2007: Basel, Switzerland - University of Basel
- 2006: Lund, Sweden - Department of Experimental Medicine, Lund University
- 2005: Xi'an, China - Invited talk: Fourth Military Medical University
- 2005: La Jolla, CA, USA - Invited Seminar at Johnson and Johnson
- 2005: Pecs, Hungary - 11th Annual Congress of the Hungarian Neuroscience Society (symposium speaker)
- 2005: San Diego, USA - Mini Symposium, Department of Anesthesiology, University of California
- 2004: Kyoto, Japan - International Federation of Associations of Anatomists (IFAA)
- 2003: Torino, Italy - European Colloquium on Pain
- 2003: Prague, Czech Republic - International Brain Research Organization (IBRO) Satellite symposium
- 2002: Tubingen, Germany - German Physiological Society, Symposium speaker
- 2002: San Diego, CA, USA - IASP 10th World Congress, Symposium speaker
- 2001: Montreal, Canada - University of Montreal, Peptide Receptors.
- 2001: Bristol, UK - Physiological Society, Symposium speaker
Professional Learned Society
- 2011 - present: Society of Biology - Fellow
- 2006 - present: Physiological Society - Member
- 2001 - present: International Association for the Study of Pain - Member
- 1997 - present: British Neuroscience Association - Member
- 1997 - present: Society for Neuroscience - Member
- 1996 - present: European Neuroscience Association - Member


